Crypt-Villus Scaffold Architecture for Bioengineering Functional Human Intestinal Epithelium
- PMID: 36191009
- PMCID: PMC10379436
- DOI: 10.1021/acsbiomaterials.2c00851
Crypt-Villus Scaffold Architecture for Bioengineering Functional Human Intestinal Epithelium
Abstract
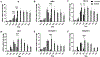
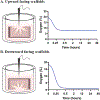
Crypt-villus architecture in the small intestine is crucial for the structural integrity of the intestinal epithelium and maintenance of gut homeostasis. We utilized three-dimensional (3D) printing and inverse molding techniques to form three-dimensional (3D) spongy scaffold systems that resemble the intestinal crypt-villus microarchitecture. The scaffolds consist of silk fibroin protein with curved lumens with rows of protruding villi with invaginating crypts to generate the architecture. Intestinal cell (Caco-2, HT29-MTX) attachment and growth, as well as long-term culture support were demonstrated with cell polarization and tissue barrier properties compared to two-dimensional (2D) Transwell culture controls. Further, physiologically relevant oxygen gradients were generated in the 3D system. The various advantages of this system may be ascribed to the more physiologically relevant 3D environment, offering a system for the exploration of disease pathogenesis, host-microbiome interactions, and therapeutic discovery.
Keywords: 3D printing; crypts; intestine tissue; oxygen profile; silk; tissue engineering; villi.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
-
- Halm D; Halm S Secretagogue response of goblet cells and columnar cells in human colonic crypts, American journal of physiology. Cell Physiol. 2000, 278, C212–C233. - PubMed
-
- Castaño AG; Garcia-Diaz M; Torras N; Altay G; Comelles J; Martinez E Dynamic photopolymerization produces complex microstructures on hydrogels in a moldless approach to generate a 3D intestinal tissue model. Biofabrication 2019, 11, No. 025007. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
