Role of Carbon Monoxide in Oxidative Stress-Induced Senescence in Human Bronchial Epithelium
- PMID: 36193088
- PMCID: PMC9526622
- DOI: 10.1155/2022/5199572
Role of Carbon Monoxide in Oxidative Stress-Induced Senescence in Human Bronchial Epithelium
Abstract
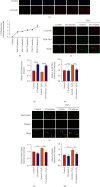
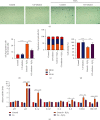
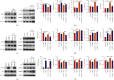
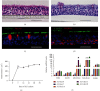
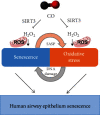
Prolonged or excessive stimulation from inhaled toxins may cause oxidative stress and DNA damage that can lead to stress-induced senescence in epithelial cells, which can contribute to several airway diseases. Mounting evidence has shown carbon monoxide (CO) confers cytoprotective effects. We investigated the effects of CO on oxidative stress-induced senescence in human airway epithelium and elucidated the underlying molecular mechanisms. Here, CO pretreatment reduced H2O2-mediated increases in total reactive oxygen species (ROS) production and mitochondrial superoxide in a human bronchial epithelial cell line (BEAS-2B). H2O2 treatment triggered a premature senescence-like phenotype with enlarged and flattened cell morphology accompanied by increased SA-β-gal activity, cell cycle arrest in G0/G1, reduced cell viability, and increased transcription of senescence-associated secretory phenotype (SASP) genes. Additionally, exposure to H2O2 increased protein levels of cellular senescence markers (p53 and p21), reduced Sirtuin 3 (SIRT3) and manganese superoxide dismutase (MnSOD) levels, and increased p53 K382 acetylation. These H2O2-mediated effects were attenuated by pretreatment with a CO-containing solution. SIRT3 silencing induced mitochondrial superoxide production and triggered a senescence-like phenotype, whereas overexpression decreased mitochondrial superoxide production and alleviated the senescence-like phenotype. Air-liquid interface (ALI) culture of primary human bronchial cells, which becomes a fully differentiated pseudostratified mucociliary epithelium, was used as a model. We found that apical and basolateral exposure to H2O2 induced a vacuolated structure that impaired the integrity of ALI cultures, increased goblet cell numbers, decreased SCGB1A1+ club cell numbers, increased p21 protein levels, and increased SASP gene transcription, consistent with our observations in BEAS-2B cells. These effects were attenuated in the apical presence of a CO-containing solution. In summary, we revealed that CO has a pivotal role in epithelial senescence by regulating ROS production via the SIRT3/MnSOD/p53/p21 pathway. This may have important implications in the prevention and treatment of age-associated respiratory pathologies.
Copyright © 2022 Meng-yun Cai et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures


Similar articles
-
Fullerenol protects retinal pigment epithelial cells from oxidative stress-induced premature senescence via activating SIRT1.Invest Ophthalmol Vis Sci. 2014 May 20;55(7):4628-38. doi: 10.1167/iovs.13-13732. Invest Ophthalmol Vis Sci. 2014. PMID: 24845634
-
A Newly Synthesized Rhamnoside Derivative Alleviates Alzheimer's Amyloid-β-Induced Oxidative Stress, Mitochondrial Dysfunction, and Cell Senescence through Upregulating SIRT3.Oxid Med Cell Longev. 2020 Feb 13;2020:7698560. doi: 10.1155/2020/7698560. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32104538 Free PMC article.
-
FAM13A regulates cellular senescence marker p21 and mitochondrial reactive oxygen species production in airway epithelial cells.Am J Physiol Lung Cell Mol Physiol. 2023 Oct 1;325(4):L460-L466. doi: 10.1152/ajplung.00141.2023. Epub 2023 Aug 22. Am J Physiol Lung Cell Mol Physiol. 2023. PMID: 37605846
-
Manganese Superoxide Dismutase Acetylation and Dysregulation, Due to Loss of SIRT3 Activity, Promote a Luminal B-Like Breast Carcinogenic-Permissive Phenotype.Antioxid Redox Signal. 2016 Aug 20;25(6):326-36. doi: 10.1089/ars.2016.6641. Epub 2016 Apr 15. Antioxid Redox Signal. 2016. PMID: 26935174 Free PMC article. Review.
-
Mycotoxins and cellular senescence: the impact of oxidative stress, hypoxia, and immunosuppression.Arch Toxicol. 2023 Feb;97(2):393-404. doi: 10.1007/s00204-022-03423-x. Epub 2022 Nov 25. Arch Toxicol. 2023. PMID: 36434400 Review.
Cited by
-
Exhaled Biomarkers for Point-of-Care Diagnosis: Recent Advances and New Challenges in Breathomics.Micromachines (Basel). 2023 Feb 4;14(2):391. doi: 10.3390/mi14020391. Micromachines (Basel). 2023. PMID: 36838091 Free PMC article. Review.
-
circ-0001454 alleviates asthma airway inflammation and remodeling via sponging miR-770-5p and regulating cbl-b.Front Cell Dev Biol. 2025 Apr 8;13:1566223. doi: 10.3389/fcell.2025.1566223. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40264707 Free PMC article.
-
Single-cell RNA sequencing reveals heterogeneity of ALI model and epithelial cell alterations after exposure to electronic cigarette aerosol.Heliyon. 2024 Sep 26;10(19):e38552. doi: 10.1016/j.heliyon.2024.e38552. eCollection 2024 Oct 15. Heliyon. 2024. PMID: 39397927 Free PMC article.
-
Effect of Electronic Cigarette Vapour Exposure on Ca2+- and cAMP-Dependent Ion Transport in Human Airway Epithelial Cells.Lung. 2025 Mar 18;203(1):48. doi: 10.1007/s00408-025-00805-7. Lung. 2025. PMID: 40100414 Free PMC article.
References
-
- Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell . 2005;120(4):513–522. - PubMed
-
- Rennard S. I. Inflammation and repair processes in chronic obstructive pulmonary disease. American Journal of Respiratory and Critical Care Medicine . 1999;160, 5 Part 2:S12–S16. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

