A neuronal action of sirtuin 1 suppresses bone mass in young and aging mice
- PMID: 36194488
- PMCID: PMC9711874
- DOI: 10.1172/JCI152868
A neuronal action of sirtuin 1 suppresses bone mass in young and aging mice
Abstract
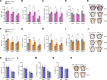
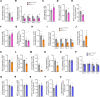
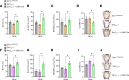
The various functions of the skeleton are influenced by extracellular cues, hormones, and neurotransmitters. One type of neuronal regulation favors bone mass accrual by inhibiting sympathetic nervous system (SNS) activity. This observation raises questions about the transcriptional mechanisms regulating catecholamine synthesis. Using a combination of genetic and pharmacological studies, we found that the histone deacetylase sirtuin 1 (SIRT1) is a transcriptional modulator of the neuronal control of bone mass. Neuronal SIRT1 reduced bone mass by increasing SNS signaling. SIRT1 did so by increasing expression of monoamine oxidase A (MAO-A), a SIRT1 target that reduces brain serotonin levels by inducing its catabolism and by suppressing tryptophan hydroxylase 2 (Tph2) expression and serotonin synthesis in the brain stem. SIRT1 upregulated brain catecholamine synthesis indirectly through serotonin, but did not directly affect dopamine β hydroxylase (Dbh) expression in the locus coeruleus. These results help us to understand skeletal changes associated with selective serotonin reuptake inhibitors (SSRIs) and may have implications for treating skeletal and metabolic diseases.
Keywords: Bone Biology; Mouse models; Neuroendocrine regulation; Neuroscience; Osteoporosis.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

