Prevalence of inverse psoriasis subtype with immune checkpoint inhibitors
- PMID: 36196370
- PMCID: PMC9525015
- DOI: 10.1093/immadv/ltac016
Prevalence of inverse psoriasis subtype with immune checkpoint inhibitors
Abstract
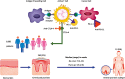
Background: Cutaneous immune-related adverse events (irAEs) are the most common irAEs caused by immune-checkpoint inhibitors (ICI). Psoriasiform eruptions, both de novo and flares, may occur. Evidence is lacking on inverse psoriasis subtype.
Methods: A retrospective study was conducted at Dana-Farber Cancer Institute/Mass General Brigham through February 2020 using databases. Confirmed inverse psoriasis cases pre-/post-ICI initiation either independently or in conjunction with other psoriasis subtypes were included. Known psoriasis cases without flare post-ICI were excluded.
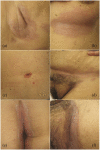
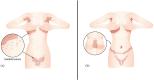
Results: A total of 262 (3%) individuals with any ICI-mediated psoriasiform cutaneous irAE were identified out of the 8683 DFCI ICI-treated patients. Of these, 13 (5% of psoriasis patients) had inverse psoriasis (mean age 68.7 years; 7/13 male sex). Median (range) time from ICI initiation to inverse psoriasis development or flare was 7 (4-12) and 3.5 (2-6) weeks, respectively. Pruritus occurred in 12/13 (92.30%) cases. 11 (85%) had inguinal involvement; other sites included gluteal cleft (6; 46%), inframammary (3; 23%), perianal (2; 15%), axilla (2; 15%), umbilicus (2; 15%), and infra-abdominal folds (1; 8%). Most (9/13) individuals had more than one site involved. The Common Terminology Criteria for Adverse Events severity was 1 in 10 (76.92%) individuals and 2 in 3 (15.38%) individuals. Six (46.15%) patients were treated initially by oncology with topical (nystatin, econazole, or clotrimazole) or systemic antifungals (fluconazole) for median (range) of 3.5 (1-7) months without improvement, for presumed candida intertrigo.
Conclusion: Patients on ICI may develop inverse psoriasis, which may be initially confused for fungal intertrigo. Delayed diagnosis can prolong symptoms, while patients are treated ineffectively with topical/systemic antifungals for presumed candida infection. Oncologist and dermatologist awareness is important to improve diagnosis of ICI-mediated inverse psoriasis, its management and affected patients' quality of life.
Keywords: flexural psoriasis; immune checkpoint inhibitors; immune-related adverse events; intertriginous psoriasis; inverse psoriasis; skin toxicity.
© The Author(s) 2022. Published by Oxford University Press on behalf of the British Society for Immunology.
Figures
References
-
- Wang G, Li C, Gao Tet al. . Clinical analysis of 48 cases of inverse psoriasis: a hospital-based study. Eur J Dermatol 2005; 15(3):176–8. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
