A randomized controlled pilot study of intranasal lidocaine in acute management of paediatric migraine and migraine-like headache
- PMID: 36200101
- PMCID: PMC9528783
- DOI: 10.1093/pch/pxac054
A randomized controlled pilot study of intranasal lidocaine in acute management of paediatric migraine and migraine-like headache
Abstract
Objectives: This study was aimed to determine the sample size required to conduct an efficacy randomized controlled trial (RCT) to evaluate superiority of intranasal (IN) lidocaine to placebo as an analgesic option for children presenting to the paediatric emergency department (PED) with migraine or posttraumatic headache with migraine features and to evaluate study protocol feasibility.
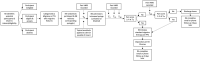
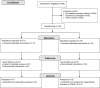
Study design: This study is a double-blind RCT of children aged 7 to 16 years at a single-centre PED. Thirty-two participants were randomized to receive either IN 2% lidocaine or 0.9% sodium chloride. The primary outcome measure was the proportion of subjects with a Verbal Numeric Rating Scale pain score of <4 at 30 and 60 minutes post-IN therapy. Primary outcome data were analyzed using a test of differences between proportions. Secondary objectives included assessing the feasibility of our study protocol by evaluating recruitment rates, adverse drug events, and PED length of stay (LOS).
Results: Six of 17 participants in the lidocaine group and 2 of 15 in the placebo group were treated successfully. Using these proportions with 95% confidence intervals and 80% power, the sample size required to find a significant difference between proportions would be 67 participants per arm. Our enrolment rate was 55% and there were no serious adverse drug events. The median PED LOS was similar between groups.
Conclusion: We determined the sample size required to conduct a definitive RCT to evaluate the superiority of IN lidocaine to placebo and found the study protocol is feasible but identified important considerations in PED migraine trial design.
Keywords: Emergency; Headache; Intranasal; Lidocaine; Migraine.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Canadian Paediatric Society. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
References
-
- Schobitz E, Qureshi F, Lewis D.. Pediatric headaches in the emergency department. Curr Pain Headache Rep 2006;10(5):391–6. - PubMed
-
- Conicella E, Raucci U, Vanacore N, et al. . The child with headache in a pediatric emergency department. Headache 2008;48(7):1005–11. - PubMed
-
- Sheridan DC, Spiro DM, Meckler GD.. Pediatric migraine: Abortive management in the emergency department. Headache 2014;54(2):235–45. - PubMed
-
- Cummings EA, Reid GJ, Finley GA, McGrath PJ, Ritchie JA.. Prevalence and source of pain in pediatric inpatients. Pain 1996;68(1):25–31. - PubMed
-
- Robbins MS, Robertson CE, Kaplan E, et al. . The sphenopalatine ganglion: anatomy, pathophysiology, and therapeutic targeting in headache. Headache 2016;56:240–58. - PubMed
LinkOut - more resources
Full Text Sources