Human embryonic stem cells cultured on hydrogels grafted with extracellular matrix protein-derived peptides with polyethylene glycol joint nanosegments
- PMID: 36200801
- PMCID: PMC9667744
- DOI: 10.1049/nbt2.12091
Human embryonic stem cells cultured on hydrogels grafted with extracellular matrix protein-derived peptides with polyethylene glycol joint nanosegments
Abstract
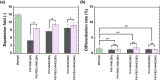
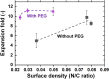
Human pluripotent stem cells (hPSCs) can be proliferated on completely synthetic materials under xeno-free cultivation conditions using biomaterials grafted with extracellular matrix protein (ECM)-derived peptides. However, cell culture biomaterials grafted with ECM-derived peptides must be prepared using a high concentration of peptide reaction solution (e.g. 1000 μg/ml), whereas the ECM concentration of the ECM-coated surface for hPSC culture is typically 5 μg/ml. We designed a polyethylene glycol (PEG) joint nanosegment (linker) to be used between base cell culture biomaterials and bioactive ECM-derived peptides to enhance the probability of contact between ECM-derived peptides and cell binding receptors of hPSCs. Vitronectin-derived peptides with glycine joint nanosegments (GCGG) were conjugated onto poly (vinyl alcohol-co-itaconic acid) hydrogels via PEG joint nanosegments, and human embryonic stem cells (hESCs) were cultivated on these hydrogels. hESCs could successfully be cultivated on hydrogels while maintaining their pluripotency and differentiation potential to differentiate into cells that are induced from three germ layers in vitro and in vivo, where only a 50 μg/ml ECM-derived peptide concentration was used when the PEG joint nanosegments were introduced into peptides that were grafted onto hydrogel surfaces. The joint nanosegments between bioactive peptides and base cell culture biomaterials were found to contribute to efficient hESC attachment and proliferation.
© 2022 The Authors. IET Nanobiotechnology published by John Wiley & Sons Ltd on behalf of The Institution of Engineering and Technology.
Conflict of interest statement
The authors report no conflict of interest in this work.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

