A novel glucagon analog with an extended half-life, HM15136, normalizes glucose levels in rodent models of congenital hyperinsulinism
- PMID: 36202918
- PMCID: PMC9537296
- DOI: 10.1038/s41598-022-21251-y
A novel glucagon analog with an extended half-life, HM15136, normalizes glucose levels in rodent models of congenital hyperinsulinism
Abstract
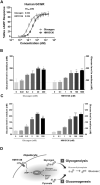
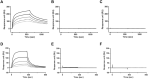
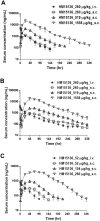
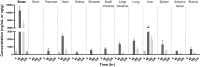
Congenital hyperinsulinism (CHI) is a rare genetic condition characterized by uncontrolled insulin secretion, resulting in hypoglycemia. Although glucagon has lately been regarded as a therapeutic option for CHI, its use is severely hampered by its poor solubility and stability at physiological pH, as well as its short duration of action. To address these constraints, we developed HM15136, a novel long-acting glucagon analog composed of a glucagon analog conjugated to the Fc fragment of human immunoglobulin G4 via a polyethylene glycol linker. In this study, we established that HM15136 was more soluble than natural glucagon (≥ 150 mg/mL vs 0.03 mg/mL). Next, we confirmed that HM15136 activated glucagon receptor in vitro and induced glycogenolysis and gluconeogenesis in rat primary hepatocytes. Pharmacokinetics (PK)/Pharmacodynamics (PD) analysis of HM15136 shows that HM15136 has a markedly longer half-life (36 h vs. < 5 min) and increased bioavailability (90%) compared to native glucagon in mice. Further, HM15136 could effectively reverse acute hypoglycemia induced by insulin challenge, and multiple doses of HM15136 could sustain increased blood glucose levels in CHI rats. In conclusion, our findings indicate that HM15136 promotes sustained elevation of blood glucose, demonstrating the potential for development as a once-weekly therapy for CHI.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

