Membrane attack complexes, endothelial cell activation, and direct allorecognition
- PMID: 36211400
- PMCID: PMC9539657
- DOI: 10.3389/fimmu.2022.1020889
Membrane attack complexes, endothelial cell activation, and direct allorecognition
Abstract
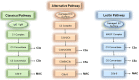
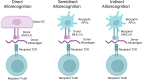
Endothelial cells (ECs) form a critical immune interface regulating both the activation and trafficking of alloreactive T cells. In the setting of solid organ transplantation, donor-derived ECs represent sites where alloreactive T cells encounter major and minor tissue-derived alloantigens. During this initial encounter, ECs may formatively modulate effector responses of these T cells through expression of inflammatory mediators. Direct allorecognition is a process whereby recipient T cells recognize alloantigen in the context of donor EC-derived HLA molecules. Direct alloresponses are strongly modulated by human ECs and are galvanized by EC-derived inflammatory mediators. Complement are immune proteins that mark damaged or foreign surfaces for immune cell activation. Following labeling by natural IgM during ischemia reperfusion injury (IRI) or IgG during antibody-mediated rejection (ABMR), the complement cascade is terminally activated in the vicinity of donor-derived ECs to locally generate the solid-phase inflammatory mediator, the membrane attack complex (MAC). Via upregulation of leukocyte adhesion molecules, costimulatory molecules, and cytokine trans-presentation, MAC strengthen EC:T cell direct alloresponses and qualitatively shape the alloimmune T cell response. These processes together promote T cell-mediated inflammation during solid organ transplant rejection. In this review we describe molecular pathways downstream of IgM- and IgG-mediated MAC assembly on ECs in the setting of IRI and ABMR of tissue allografts, respectively. We describe work demonstrating that MAC deposition on ECs generates 'signaling endosomes' that sequester and post-translationally enhance the stability of inflammatory signaling molecules to promote EC activation, a process potentiating EC-mediated direct allorecognition. Additionally, with consideration to first-in-human xenotransplantation procedures, we describe clinical therapeutics based on inhibition of the complement pathway. The complement cascade critically mediates EC activation and improved understanding of relevant effector pathways will uncover druggable targets to obviate dysregulated alloimmune T cell infiltration into tissue allografts.
Keywords: allorecognition; antibody-mediated rejection; complement; endothelial cell; transplant.
Copyright © 2022 Song, Wang, Barkestani, Mullan, Fan, Jiang, Jiang, Li and Jane-wit.
Conflict of interest statement
Work supporting development of humanized mouse models was supported in part through a research grant from Alexion Pharmaceuticals (JP). Work supporting identification of ZFYVE21 was supported in part through a research grant from Abbvie Pharmaceutical Research and Development (JP, DJ). The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
T cell Allorecognition Pathways in Solid Organ Transplantation.Front Immunol. 2018 Nov 5;9:2548. doi: 10.3389/fimmu.2018.02548. eCollection 2018. Front Immunol. 2018. PMID: 30455697 Free PMC article. Review.
-
Complement Membrane Attack Complexes Assemble NLRP3 Inflammasomes Triggering IL-1 Activation of IFN-γ-Primed Human Endothelium.Circ Res. 2019 Jun 7;124(12):1747-1759. doi: 10.1161/CIRCRESAHA.119.314845. Epub 2019 Apr 22. Circ Res. 2019. PMID: 31170059 Free PMC article.
-
Effects of antibody reactivity to major histocompatibility complex (MHC) and non-MHC alloantigens on graft endothelial cells in heart allograft rejection.Transplantation. 2000 May 15;69(9):1899-906. doi: 10.1097/00007890-200005150-00027. Transplantation. 2000. PMID: 10830229
-
Propagation and characterization of lymphocytes from transplant biopsies.Crit Rev Immunol. 1991;10(6):455-80. Crit Rev Immunol. 1991. PMID: 1830745 Review.
-
Extracellular Vesicles as Mediators of Cellular Crosstalk Between Immune System and Kidney Graft.Front Immunol. 2020 Feb 27;11:74. doi: 10.3389/fimmu.2020.00074. eCollection 2020. Front Immunol. 2020. PMID: 32180768 Free PMC article. Review.
Cited by
-
Mechanome-Guided Strategies in Regenerative Rehabilitation.Curr Opin Biomed Eng. 2024 Mar;29:100516. doi: 10.1016/j.cobme.2023.100516. Epub 2023 Nov 30. Curr Opin Biomed Eng. 2024. PMID: 38586151 Free PMC article.
-
Peripheral blood transcriptomic analysis identifies potential inflammation and immune signatures for central retinal artery occlusion.Sci Rep. 2024 Mar 28;14(1):7398. doi: 10.1038/s41598-024-57052-8. Sci Rep. 2024. PMID: 38548806 Free PMC article.
-
Immune profiling of kidney transplant recipients: the role of Tribbles-1, Perforin, and Granzyme B in chronic humoral rejection.Mol Biol Rep. 2025 Jun 9;52(1):573. doi: 10.1007/s11033-025-10688-9. Mol Biol Rep. 2025. PMID: 40488991
-
Hedgehog costimulation during ischemia-reperfusion injury potentiates cytokine and homing responses of CD4+ T cells.Front Immunol. 2023 Oct 17;14:1248027. doi: 10.3389/fimmu.2023.1248027. eCollection 2023. Front Immunol. 2023. PMID: 37915586 Free PMC article.
-
Deciphering the Cell-Specific Effect of Osteoblast-Macrophage Crosstalk in Periodontitis.Tissue Eng Part A. 2023 Nov;29(21-22):579-593. doi: 10.1089/ten.TEA.2023.0104. Epub 2023 Oct 6. Tissue Eng Part A. 2023. PMID: 37639358 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

