Combined effects of Bacillus sp. M6 strain and Sedum alfredii on rhizosphere community and bioremediation of cadmium polluted soils
- PMID: 36212314
- PMCID: PMC9533712
- DOI: 10.3389/fpls.2022.913787
Combined effects of Bacillus sp. M6 strain and Sedum alfredii on rhizosphere community and bioremediation of cadmium polluted soils
Abstract
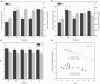
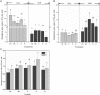
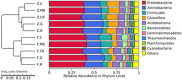
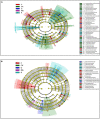
Concerns regarding inevitable soil translocation and bioaccumulation of cadmium (Cd) in plants have been escalating in concomitance with the posed phytotoxicity and threat to human health. Exhibiting a Cd tolerance, Bacillus sp. M6 strain has been reported as a soil amendment owing to its capability of reducing metal bioavailability in soils. The present study investigated the rhizospheric bacterial community of the Cd hyperaccumulator Sedum alfredii using 16S rRNA gene sequencing. Additionally, the Cd removal efficiency of strain Bacillus sp. M6 was enhanced by supplementing with biochar (C), glutamic acid (G), and rhamnolipid (R) to promote the phytoremediation effect of hyperaccumulator S. alfredii. To the best of our knowledge, this is the first time the amendments such as C, G, and R together with the plant-microbe system S. alfredii-Bacillus sp. M6 has been used for Cd bioremediation. The results showed that soil CaCl2 and DTPA (Diethylenetriamine penta-acetic acid) extractable Cd increased by 52.77 and 95.08%, respectively, in all M6 treatments compared to unamended control (CK). Sedum alfredii with Bacillus sp. M6 supplemented with biochar and rhamnolipid displayed a higher phytoremediation effect, and the removal capability of soil Cd (II) reached up to 16.47%. Moreover, remediation of Cd polluted soil by Bacillus sp. M6 also had an impact on the soil microbiome, including ammonia-oxidizing bacteria (AOB), ammonia-oxidizing archaea (AOA), and cadmium transporting ATPase (cadA) genes. Quantitative PCR analysis confirmed the Bacillus sp. M6 strain increased the abundance of AOB and cadA in both low Cd (LC) and high Cd (HC) soils compared to AOA gene abundance. Besides, the abundance of Proteobacteria and Actinobacteria was found to be highest in both soils representing high tolerance capacity against Cd. While Firmicutes ranked third, indicating that the additionof strain could not make it the most dominant species. The results suggested the presence of the hyperaccumulator S. alfredii and Cd tolerant strain Bacillus sp. M6 supplemented with biochar, and rhamnolipid, play a unique and essential role in the remediation process and reducing the bioavailability of Cd.
Keywords: Bacillus sp. M6; Sedum alfredii; biochar (BC); bioremediation; cadmium; rhamnolipid; soil microbial community.
Copyright © 2022 Abid, Zhang, He, Wang, Batool, Di and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Cadmium Exposure-Sedum alfredii Planting Interactions Shape the Bacterial Community in the Hyperaccumulator Plant Rhizosphere.Appl Environ Microbiol. 2018 May 31;84(12):e02797-17. doi: 10.1128/AEM.02797-17. Print 2018 Jun 15. Appl Environ Microbiol. 2018. PMID: 29654182 Free PMC article.
-
Inoculation of multi-metal-resistant Bacillus sp. to a hyperaccumulator plant Sedum alfredii for facilitating phytoextraction of heavy metals from contaminated soil.Chemosphere. 2024 Oct;366:143464. doi: 10.1016/j.chemosphere.2024.143464. Epub 2024 Oct 3. Chemosphere. 2024. PMID: 39368497
-
Appropriate sulfur fertilization in contaminated soil enhanced the cadmium uptake by hyperaccumulator Sedum alfredii Hance.Ecotoxicol Environ Saf. 2024 Sep 15;283:116870. doi: 10.1016/j.ecoenv.2024.116870. Epub 2024 Aug 13. Ecotoxicol Environ Saf. 2024. PMID: 39137467
-
Biochar loaded with bacteria enhanced Cd/Zn phytoextraction by facilitating plant growth and shaping rhizospheric microbial community.Environ Pollut. 2023 Jun 15;327:121559. doi: 10.1016/j.envpol.2023.121559. Epub 2023 Apr 4. Environ Pollut. 2023. PMID: 37023890
-
Cadmium Phytotoxicity, Tolerance, and Advanced Remediation Approaches in Agricultural Soils; A Comprehensive Review.Front Plant Sci. 2022 Mar 9;13:773815. doi: 10.3389/fpls.2022.773815. eCollection 2022. Front Plant Sci. 2022. PMID: 35371142 Free PMC article. Review.
Cited by
-
The Ecological Restoration Strategies in Terrestrial Ecosystems Were Reviewed: A New Trend Based on Soil Microbiomics.Ecol Evol. 2025 Mar 6;15(3):e70994. doi: 10.1002/ece3.70994. eCollection 2025 Mar. Ecol Evol. 2025. PMID: 40060716 Free PMC article. Review.
References
-
- Argun M. E., Dursun S., Karatas M. (2009). Removal of Cd (II) and Pb (II), Cu (II) and Ni (II) from water using modified pine bark. Desalination 24 519–527. 10.1016/j.desal.2009.01.020 - DOI
-
- Atkinson C. J., Fitzgerald J. D., Hipps N. A. (2010). Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337 1–18. 10.1007/s11104-010-0464-5 - DOI
LinkOut - more resources
Full Text Sources
Research Materials

