15-Deoxy-Delta-12,14-prostaglandin J2 modulates pro-labour and pro-inflammatory responses in human myocytes, vaginal and amnion epithelial cells
- PMID: 36213265
- PMCID: PMC9533017
- DOI: 10.3389/fendo.2022.983924
15-Deoxy-Delta-12,14-prostaglandin J2 modulates pro-labour and pro-inflammatory responses in human myocytes, vaginal and amnion epithelial cells
Abstract
Background: Prematurity is the leading cause of childhood death under the age of five. The aetiology of preterm birth is multifactorial; however, inflammation and infection are the most common causal factors, supporting a potential role for immunomodulation as a therapeutic strategy. 15-Deoxy-Delta-12,14-prostaglandin J2 (15dPGJ2) is an anti-inflammatory prostaglandin and has been shown to delay lipopolysaccharide (LPS) induced preterm labour in mice and improve pup survival. This study explores the immunomodulatory effect of 15dPGJ2 on the transcription factors NF-κB and AP-1, pro-inflammatory cytokines, and contraction associated proteins in human cultured myocytes, vaginal epithelial cell line (VECs) and primary amnion epithelial cells (AECs).
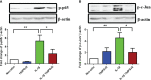
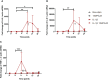
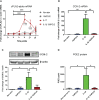
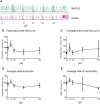
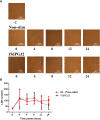
Methods: Cells were pre-incubated with 32µM of 15dPGJ2 and stimulated with 1ng/mL of IL-1β as an in vitro model of inflammation. Western immunoblotting was used to detect phosphorylated p-65 and phosphorylated c-Jun as markers of NF-κB and AP-1 activation, respectively. mRNA expression of the pro-inflammatory cytokines IL-6, IL-8, and TNF-α was examined, and protein expression of COX-2 and PGE2 were detected by western immunoblotting and ELISA respectively. Myometrial contractility was examined ex-vivo using a myograph.
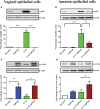
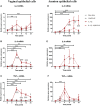
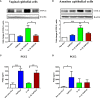
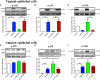
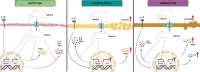
Results: 15dPGJ2 inhibited IL-1β-induced activation of NF-κB and AP-1, and expression of IL-6, IL-8, TNF-α, COX-2 and PGE2 in myocytes, with no effect on myometrial contractility or cell viability. Despite inhibiting IL-1β-induced activation of NF-κB, expression of IL-6, TNF-α, and COX-2, 15dPGJ2 led to activation of AP-1, increased production of PGE2 and increased cell death in VECs and AECs.
Conclusion: We conclude that 15dPGJ2 has differential effects on inflammatory modulation depending on cell type and is therefore unlikely to be a useful therapeutic agent for the prevention of preterm birth.
Keywords: 15dPGJ2; activator protein (AP)-1; cytokines; inflammation; nuclear factor - kappa B (NF - κB); preterm labour (PTL); prostaglandins.
Copyright © 2022 Rasheed, Lee, Kim, Teoh, MacIntyre, Bennett and Sykes.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Sulfasalazine augments a pro-inflammatory response in interleukin-1β-stimulated amniocytes and myocytes.Immunology. 2015 Dec;146(4):630-44. doi: 10.1111/imm.12534. Epub 2015 Nov 3. Immunology. 2015. PMID: 26395271 Free PMC article.
-
Propofol Suppresses LPS-Induced Inflammation in Amnion Cells via Inhibition of NF-κB Activation.Tissue Eng Regen Med. 2019 May 29;16(3):301-309. doi: 10.1007/s13770-019-00194-y. eCollection 2019 Jun. Tissue Eng Regen Med. 2019. PMID: 31205858 Free PMC article.
-
A20, an essential component of the ubiquitin-editing protein complex, is a negative regulator of inflammation in human myometrium and foetal membranes.Mol Hum Reprod. 2017 Sep 1;23(9):628-645. doi: 10.1093/molehr/gax041. Mol Hum Reprod. 2017. PMID: 28911210
-
DXXK exerts anti-inflammatory effects by inhibiting the lipopolysaccharide-induced NF-κB/COX-2 signalling pathway and the expression of inflammatory mediators.J Ethnopharmacol. 2016 Feb 3;178:199-208. doi: 10.1016/j.jep.2015.11.016. Epub 2015 Nov 10. J Ethnopharmacol. 2016. PMID: 26571085
-
Targeting inflammatory pathways for tumor radiosensitization.Biochem Pharmacol. 2010 Dec 15;80(12):1904-14. doi: 10.1016/j.bcp.2010.06.039. Epub 2010 Jun 30. Biochem Pharmacol. 2010. PMID: 20599771 Free PMC article. Review.
Cited by
-
TLR signaling pathway and the effects of main immune cells and epigenetics factors on the diagnosis and treatment of infertility and sterility.Heliyon. 2024 Jul 26;10(15):e35345. doi: 10.1016/j.heliyon.2024.e35345. eCollection 2024 Aug 15. Heliyon. 2024. PMID: 39165943 Free PMC article. Review.
-
Meconopsis quintuplinervia Regel Improves Cutibacterium acnes-Induced Inflammatory Responses in a Mouse Ear Edema Model and Suppresses Pro-Inflammatory Chemokine Production via the MAPK and NF-κB Pathways in RAW264.7 Cells.Ann Dermatol. 2023 Dec;35(6):408-416. doi: 10.5021/ad.22.206. Ann Dermatol. 2023. PMID: 38086354 Free PMC article.
References
-
- Howson CP, Kinney MV, Lawn JE. Born too soon: The global action report on preterm birth. In: March of dimes, PMNCH, save the children. Geneva: World Health Organisation; (2012).
-
- UnitedNations . (2022). Available at: https://sdgs.un.org/goals/goal3.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

