The Effects of Middle-ear Stiffness on the Auditory Brainstem Neural Encoding of Phase
- PMID: 36214911
- PMCID: PMC9549819
- DOI: 10.1007/s10162-022-00872-0
The Effects of Middle-ear Stiffness on the Auditory Brainstem Neural Encoding of Phase
Abstract
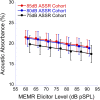
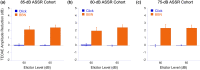
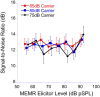
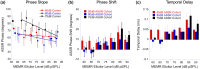
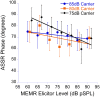
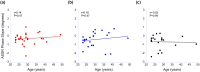
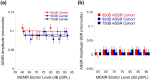
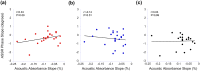
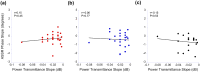
The middle-ear system relies on a balance of mass and stiffness characteristics for transmitting sound from the external environment to the cochlea and auditory neural pathway. Phase is one aspect of sound that, when transmitted and encoded by both ears, contributes to binaural cue sensitivity and spatial hearing. The study aims were (i) to investigate the effects of middle-ear stiffness on the auditory brainstem neural encoding of phase in human adults with normal pure-tone thresholds and (ii) to investigate the relationships between middle-ear stiffness-induced changes in wideband acoustic immittance and neural encoding of phase. The auditory brainstem neural encoding of phase was measured using the auditory steady-state response (ASSR) with and without middle-ear stiffness elicited via contralateral activation of the middle-ear muscle reflex (MEMR). Middle-ear stiffness was quantified using a wideband acoustic immittance assay of acoustic absorbance. Statistical analyses demonstrated decreased ASSR phase lag and decreased acoustic absorbance with contralateral activation of the MEMR, consistent with increased middle-ear stiffness changing the auditory brainstem neural encoding of phase. There were no statistically significant correlations between stiffness-induced changes in wideband acoustic absorbance and ASSR phase. The findings of this study may have important implications for understanding binaural cue sensitivity and horizontal plane sound localization in audiologic and otologic clinical populations that demonstrate changes in middle-ear stiffness, including cochlear implant recipients who use combined electric and binaural acoustic hearing and otosclerosis patients.
Keywords: auditory brainstem; auditory steady-state response; middle-ear stiffness; phase.
© 2022. The Author(s) under exclusive licence to Association for Research in Otolaryngology.
Conflict of interest statement
René H. Gifford is a consultant for Advanced Bionics, Cochlear Limited, Akouos, and Frequency Therapeutics. Linda J. Hood is a consultant for Akouos and Pfizer. These consultant roles are not in conflict with the work reported here. Rafael E. Delgado is employed by and a stock holder of Intelligent Hearing Systems. There are no other conflicts of interest, financial or otherwise, to disclose.
Figures

Similar articles
-
Identifying Otosclerosis with Aural Acoustical Tests of Absorbance, Group Delay, Acoustic Reflex Threshold, and Otoacoustic Emissions.J Am Acad Audiol. 2017 Oct;28(9):838-860. doi: 10.3766/jaaa.16172. J Am Acad Audiol. 2017. PMID: 28972472 Free PMC article.
-
Maturation of the occlusion effect: a bone conduction auditory steady state response study in infants and adults with normal hearing.Ear Hear. 2011 Nov-Dec;32(6):708-19. doi: 10.1097/AUD.0b013e31821de1b0. Ear Hear. 2011. PMID: 21617531 Clinical Trial.
-
[New hearing test for functional deafness--the procedure to estimate the genuine hearing threshold].Nihon Jibiinkoka Gakkai Kaiho. 1999 Sep;102(9):1028-35. doi: 10.3950/jibiinkoka.102.1028. Nihon Jibiinkoka Gakkai Kaiho. 1999. PMID: 10554555 Clinical Trial. Japanese.
-
Acoustic reflex measurement.Ear Hear. 2013 Jul;34 Suppl 1:43S-47S. doi: 10.1097/AUD.0b013e31829c70d9. Ear Hear. 2013. PMID: 23900179 Review.
-
Detection of hearing impairment with the acoustic stapedius reflex.Ear Hear. 1993 Feb;14(1):3-10. doi: 10.1097/00003446-199302000-00002. Ear Hear. 1993. PMID: 8444335 Review.
References
-
- Merchant SN, Rosowski JJ (2003) Auditory physiology. Surg Ear 59–82
-
- Avan P, Büki B, Maat B, Dordain M, Wit HP (2000) Middle ear influence on otoacoustic emissions. I: noninvasive investigation of the human transmission apparatus and comparison with model results. Hear Res 140(1–2):189–201 - PubMed
-
- Büki B, Wit HP, Avan P (2000) Olivocochlear efferent vs. middle-ear contributions to the alteration of otoacoustic emissions by contralateral noise. Brain Res 852(1):140–150 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

