Mitochondrial network expansion and dynamic redistribution during islet morphogenesis in zebrafish larvae
- PMID: 36217213
- PMCID: PMC10092693
- DOI: 10.1002/1873-3468.14508
Mitochondrial network expansion and dynamic redistribution during islet morphogenesis in zebrafish larvae
Abstract
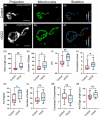
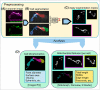
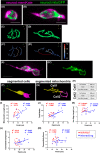
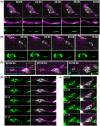
Mitochondria, organelles critical for energy production, modify their shape and location in response to developmental state and metabolic demands. Mitochondria are altered in diabetes, but the mechanistic basis is poorly defined, due to difficulties in assessing mitochondria within an intact organism. Here, we use in vivo imaging in transparent zebrafish larvae to demonstrate filamentous, interconnected mitochondrial networks within islet cells. Mitochondrial movements highly resemble what has been reported for human islet cells in vitro, showing conservation in behaviour across species and cellular context. During islet development, mitochondrial content increases with emergence of cell motility, and mitochondria disperse within fine protrusions. Overall, this work presents quantitative analysis of mitochondria within their native environment and provides insights into mitochondrial behaviour during organogenesis.
Keywords: image analysis; in vivo imaging; islet; mitochondria; pancreas; zebrafish.
© 2022 The Authors. FEBS Letters published by John Wiley & Sons Ltd on behalf of Federation of European Biochemical Societies.
Figures

Similar articles
-
In vivo imaging of emerging endocrine cells reveals a requirement for PI3K-regulated motility in pancreatic islet morphogenesis.Development. 2018 Feb 8;145(3):dev158477. doi: 10.1242/dev.158477. Development. 2018. PMID: 29386244 Free PMC article.
-
A bioenergetics assay for studying the effects of environmental stressors on mitochondrial function in vivo in zebrafish larvae.Comp Biochem Physiol C Toxicol Pharmacol. 2017 Feb;192:23-32. doi: 10.1016/j.cbpc.2016.12.001. Epub 2016 Dec 7. Comp Biochem Physiol C Toxicol Pharmacol. 2017. PMID: 27939721 Free PMC article.
-
Neutrophil-specific knockout demonstrates a role for mitochondria in regulating neutrophil motility in zebrafish.Dis Model Mech. 2018 Mar 28;11(3):dmm033027. doi: 10.1242/dmm.033027. Dis Model Mech. 2018. PMID: 29590639 Free PMC article.
-
Mitochondria as targets for toxicity and metabolism research using zebrafish.Biochim Biophys Acta Gen Subj. 2020 Aug;1864(8):129634. doi: 10.1016/j.bbagen.2020.129634. Epub 2020 May 15. Biochim Biophys Acta Gen Subj. 2020. PMID: 32417171 Review.
-
Mitochondria on guard: role of mitochondrial fusion and fission in the regulation of apoptosis.Adv Exp Med Biol. 2010;687:131-42. doi: 10.1007/978-1-4419-6706-0_8. Adv Exp Med Biol. 2010. PMID: 20919642 Review.
References
-
- Xu X, Duan S, Yi F, Ocampo A, Liu GH, Izpisua Belmonte JC. Mitochondrial regulation in pluripotent stem cells. Cell Metab. 2013;18:325–32. - PubMed
-
- Supale S, Li N, Brun T, Maechler P. Mitochondrial dysfunction in pancreatic beta cells. Trends Endocrinol Metab. 2012;23:477–87. - PubMed
-
- Westermann B. Bioenergetic role of mitochondrial fusion and fission. Biochim Biophys Acta. 2012;1817:1833–8. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

