Lysosomal solute and water transport
- PMID: 36219209
- PMCID: PMC9559593
- DOI: 10.1083/jcb.202109133
Lysosomal solute and water transport
Abstract
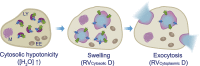
Lysosomes mediate hydrolase-catalyzed macromolecule degradation to produce building block catabolites for reuse. Lysosome function requires an osmo-sensing machinery that regulates osmolytes (ions and organic solutes) and water flux. During hypoosmotic stress or when undigested materials accumulate, lysosomes become swollen and hypo-functional. As a membranous organelle filled with cargo macromolecules, catabolites, ions, and hydrolases, the lysosome must have mechanisms that regulate its shape and size while coordinating content exchange. In this review, we discussed the mechanisms that regulate lysosomal fusion and fission as well as swelling and condensation, with a focus on solute and water transport mechanisms across lysosomal membranes. Lysosomal H+, Na+, K+, Ca2+, and Cl- channels and transporters sense trafficking and osmotic cues to regulate both solute flux and membrane trafficking. We also provide perspectives on how lysosomes may adjust the volume of themselves, the cytosol, and the cytoplasm through the control of lysosomal solute and water transport.
© 2022 Hu et al.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

