PTRH2 is Necessary for Purkinje Cell Differentiation and Survival and its Loss Recapitulates Progressive Cerebellar Atrophy and Ataxia Seen in IMNEPD Patients
- PMID: 36219306
- PMCID: PMC10657312
- DOI: 10.1007/s12311-022-01488-z
PTRH2 is Necessary for Purkinje Cell Differentiation and Survival and its Loss Recapitulates Progressive Cerebellar Atrophy and Ataxia Seen in IMNEPD Patients
Abstract
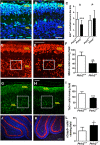
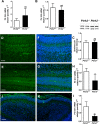
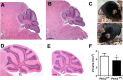
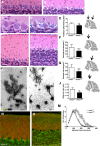
Hom ozygous variants in the peptidyl-tRNA hydrolase 2 gene (PTRH2) cause infantile-onset multisystem neurologic, endocrine, and pancreatic disease. The objective is to delineate the mechanisms underlying the core cerebellar phenotype in this disease. For this, we generated constitutive (Ptrh2LoxPxhCMVCre, Ptrh2-/- mice) and Purkinje cell (PC) specific (Ptrh2LoxPxPcp2Cre, Ptrh2ΔPCmice) Ptrh2 mutant mouse models and investigated the effect of the loss of Ptrh2 on cerebellar development. We show that Ptrh2-/- knockout mice had severe postnatal runting and lethality by postnatal day 14. Ptrh2ΔPC PC specific knockout mice survived until adult age; however, they showed progressive cerebellar atrophy and functional cerebellar deficits with abnormal gait and ataxia. PCs of Ptrh2ΔPC mice had reduced cell size and density, stunted dendrites, and lower levels of ribosomal protein S6, a readout of the mammalian target of rapamycin pathway. By adulthood, there was a marked loss of PCs. Thus, we identify a cell autonomous requirement for PTRH2 in PC maturation and survival. Loss of PTRH2 in PCs leads to downregulation of the mTOR pathway and PC atrophy. This suggests a molecular mechanism underlying the ataxia and cerebellar atrophy seen in patients with PTRH2 mutations leading to infantile-onset multisystem neurologic, endocrine, and pancreatic disease.
Keywords: Ataxia; Cell survival; IMNEPD; PTRH2; Purkinje cells; mTOR.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Phenotype variability of infantile-onset multisystem neurologic, endocrine, and pancreatic disease IMNEPD.Orphanet J Rare Dis. 2016 Apr 29;11(1):52. doi: 10.1186/s13023-016-0433-z. Orphanet J Rare Dis. 2016. PMID: 27129381 Free PMC article.
-
PTRH2 Gene Variants: Recent Review of the Phenotypic Features and Their Bioinformatics Analysis.Genes (Basel). 2023 Apr 30;14(5):1031. doi: 10.3390/genes14051031. Genes (Basel). 2023. PMID: 37239392 Free PMC article.
-
Mutations in PTRH2 cause novel infantile-onset multisystem disease with intellectual disability, microcephaly, progressive ataxia, and muscle weakness.Ann Clin Transl Neurol. 2014 Dec;1(12):1024-35. doi: 10.1002/acn3.149. Epub 2014 Dec 3. Ann Clin Transl Neurol. 2014. PMID: 25574476 Free PMC article.
-
Infantile-Onset Multisystem Neurologic, Endocrine, and Pancreatic Disease: Case and Review.Can J Neurol Sci. 2019 Jul;46(4):459-463. doi: 10.1017/cjn.2019.35. Epub 2019 May 6. Can J Neurol Sci. 2019. PMID: 31057140 Review.
-
PTRH2: an adhesion regulated molecular switch at the nexus of life, death, and differentiation.Cell Death Discov. 2020 Nov 12;6(1):124. doi: 10.1038/s41420-020-00357-0. Cell Death Discov. 2020. PMID: 33298880 Free PMC article. Review.
Cited by
-
Novel PTRH2 gene variant causing IMNEPD (infantile-onset multisystem neurologic, endocrine, and pancreatic disease) in 2 Saudi siblings.Clin Exp Pediatr. 2023 May;66(5):223-225. doi: 10.3345/cep.2022.01074. Epub 2023 Mar 23. Clin Exp Pediatr. 2023. PMID: 36949636 Free PMC article. No abstract available.
-
Identification of anoikis-related genes classification patterns and immune infiltration characterization in ischemic stroke based on machine learning.Front Aging Neurosci. 2023 Mar 23;15:1142163. doi: 10.3389/fnagi.2023.1142163. eCollection 2023. Front Aging Neurosci. 2023. PMID: 37032832 Free PMC article.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

