Adverse prognostic impact of the loss of STAG2 protein expression in patients with newly diagnosed localised Ewing sarcoma: A report from the Children's Oncology Group
- PMID: 36221002
- PMCID: PMC9726932
- DOI: 10.1038/s41416-022-01977-2
Adverse prognostic impact of the loss of STAG2 protein expression in patients with newly diagnosed localised Ewing sarcoma: A report from the Children's Oncology Group
Abstract
Background: Ewing sarcoma (EWS) is an aggressive sarcoma with no validated molecular biomarkers. We aimed to determine the frequency of STAG2 protein loss by immunohistochemistry (IHC) and whether loss of expression is associated with outcome.
Methods: We performed a retrospective cohort study of patients with EWS enrolled to Children's Oncology Group studies. We obtained unstained slides from 235 patients and DNA for sequencing from 75 patients. STAG2 expression was tested for association with clinical features and survival was estimated using Kaplan-Meier methods with log-rank tests.
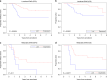
Results: In total, 155 cases passed quality control for STAG2 IHC. STAG2 expression in 20/155 cases could not be categorised with the limited available tissue, leaving 135 patients with definitive STAG2 IHC. In localised and metastatic disease, STAG2 was lost in 29/108 and 6/27 cases, respectively. Among patients with IHC and sequencing, 0/17 STAG2 expressing cases had STAG2 mutations, and 2/7 cases with STAG2 loss had STAG2 mutations. Among patients with localised disease, 5-year event-free survival was 54% (95% CI 34-70%) and 75% (95% CI 63-84%) for patients with STAG2 loss vs. expression (P = 0.0034).
Conclusion: STAG2 loss of expression is identified in a population of patients without identifiable STAG2 mutations and carries a poor prognosis.
© 2022. The Author(s).
Conflict of interest statement
DSS has received fees for consulting from Boehringer Ingelheim. KAJ has received fees for consulting, travel support or honoraria for speaking from Bayer, Roche, Foundation Medicine and Ipsen. SGD has received fees for consulting and advisory board roles from Amgen, Bayer and Jazz Pharmaceuticals and has received travel expenses from Loxo Oncology, Roche, and Salarius. KS receives grant funding as part of the DFCI/Novartis Drug Discovery Program and from Kronos Bio, is a SAB member with stock options with Auron Therapeutics, and has previously consulted for AstraZeneca. AJC has received consulting fees from Bayer. BDC has received funding for unrelated experiments from Gradalis, and his spouse was previously employed and received equity from Acceleron Pharma.
Figures


References
-
- Tirode F, Surdez D, Ma X, Parker M, Le Deley MC, Bahrami A, et al. Genomic landscape of Ewing sarcoma defines an aggressive subtype with co-association of STAG2 and TP53 mutations. Cancer Discov. 2014;4:1342–53. doi: 10.1158/2159-8290.CD-14-0622. - DOI - PMC - PubMed
-
- Crompton BD, Stewart C, Taylor-Weiner A, Alexe G, Kurek KC, Calicchio ML, et al. The genomic landscape of pediatric Ewing sarcoma. Cancer Discov. 2014;4:1326–41. doi: 10.1158/2159-8290.CD-13-1037. - DOI - PubMed
-
- Leavey PJ, Laack NN, Krailo MD, Buxton A, Randall RL, DuBois SG, et al. Phase III Trial Adding Vincristine-Topotecan-Cyclophosphamide to the Initial Treatment of Patients With Nonmetastatic Ewing Sarcoma: A Children’s Oncology Group Report. J Clin Oncol. 2021;39:4029–38. doi: 10.1200/JCO.21.00358. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

