SERINC5 restricts influenza virus infectivity
- PMID: 36223419
- PMCID: PMC9591065
- DOI: 10.1371/journal.ppat.1010907
SERINC5 restricts influenza virus infectivity
Abstract
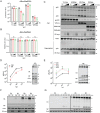
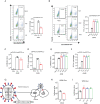
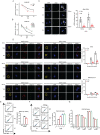
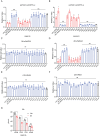
SERINC5 is a multi-span transmembrane protein that is incorporated into HIV-1 particles in producing cells and inhibits HIV-1 entry. Multiple retroviruses like HIV-1, equine infectious anemia virus and murine leukemia virus are subject to SERINC5 inhibition, while HIV-1 pseudotyped with envelope glycoproteins of vesicular stomatitis virus and Ebola virus are resistant to SERINC5. The antiviral spectrum and the underlying mechanisms of SERINC5 restriction are not completely understood. Here we show that SERINC5 inhibits influenza A virus infection by targeting virus-cell membrane fusion at an early step of infection. Further results show that different influenza hemagglutinin (HA) subtypes exhibit diverse sensitivities to SERINC5 restriction. Analysis of the amino acid sequences of influenza HA1 strains indicates that HA glycosylation sites correlate with the sensitivity of influenza HA to SERINC5, and the inhibitory effect of SERINC5 was lost when certain HA glycosylation sites were mutated. Our study not only expands the antiviral spectrum of SERINC5, but also reveals the role of viral envelope glycosylation in resisting SERINC5 restriction.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Influence of Different Glycoproteins and of the Virion Core on SERINC5 Antiviral Activity.Viruses. 2021 Jun 30;13(7):1279. doi: 10.3390/v13071279. Viruses. 2021. PMID: 34209034 Free PMC article.
-
A Conserved Acidic-Cluster Motif in SERINC5 Confers Partial Resistance to Antagonism by HIV-1 Nef.J Virol. 2020 Mar 17;94(7):e01554-19. doi: 10.1128/JVI.01554-19. Print 2020 Mar 17. J Virol. 2020. PMID: 31941773 Free PMC article.
-
Effect of HIV-1 Env on SERINC5 Antagonism.J Virol. 2017 Jan 31;91(4):e02214-16. doi: 10.1128/JVI.02214-16. Print 2017 Feb 15. J Virol. 2017. PMID: 27928004 Free PMC article.
-
SERINC as a Restriction Factor to Inhibit Viral Infectivity and the Interaction with HIV.J Immunol Res. 2017;2017:1548905. doi: 10.1155/2017/1548905. Epub 2017 Nov 22. J Immunol Res. 2017. PMID: 29359168 Free PMC article. Review.
-
SERINC5 counters retroviruses and non-retroviruses.Front Cell Infect Microbiol. 2025 Jan 20;14:1516806. doi: 10.3389/fcimb.2024.1516806. eCollection 2024. Front Cell Infect Microbiol. 2025. PMID: 39902183 Free PMC article. Review.
Cited by
-
Murine Leukemia Virus GlycoGag Antagonizes SERINC5 via ER-phagy Receptor RETREG1.bioRxiv [Preprint]. 2025 Mar 6:2025.03.06.641798. doi: 10.1101/2025.03.06.641798. bioRxiv. 2025. PMID: 40093084 Free PMC article. Preprint.
-
SERINC5 Mediates a Postintegration Block to HIV-1 Gene Expression in Macrophages.mBio. 2023 Apr 25;14(2):e0016623. doi: 10.1128/mbio.00166-23. Epub 2023 Mar 28. mBio. 2023. PMID: 36976020 Free PMC article.
-
SERINC5: One antiviral factor to bind them all.PLoS Pathog. 2023 Jan 19;19(1):e1011076. doi: 10.1371/journal.ppat.1011076. eCollection 2023 Jan. PLoS Pathog. 2023. PMID: 36656836 Free PMC article. No abstract available.
-
Host cell restriction factors of equine infectious anemia virus.Virol Sin. 2023 Aug;38(4):485-496. doi: 10.1016/j.virs.2023.07.001. Epub 2023 Jul 5. Virol Sin. 2023. PMID: 37419416 Free PMC article. Review.
-
Influenza Virus Host Restriction Factors: The ISGs and Non-ISGs.Pathogens. 2024 Jan 29;13(2):127. doi: 10.3390/pathogens13020127. Pathogens. 2024. PMID: 38392865 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

