Extra-cardiac BCAA catabolism lowers blood pressure and protects from heart failure
- PMID: 36223763
- PMCID: PMC9633425
- DOI: 10.1016/j.cmet.2022.09.008
Extra-cardiac BCAA catabolism lowers blood pressure and protects from heart failure
Abstract
Pharmacologic activation of branched-chain amino acid (BCAA) catabolism is protective in models of heart failure (HF). How protection occurs remains unclear, although a causative block in cardiac BCAA oxidation is widely assumed. Here, we use in vivo isotope infusions to show that cardiac BCAA oxidation in fact increases, rather than decreases, in HF. Moreover, cardiac-specific activation of BCAA oxidation does not protect from HF even though systemic activation does. Lowering plasma and cardiac BCAAs also fails to confer significant protection, suggesting alternative mechanisms of protection. Surprisingly, activation of BCAA catabolism lowers blood pressure (BP), a known cardioprotective mechanism. BP lowering occurred independently of nitric oxide and reflected vascular resistance to adrenergic constriction. Mendelian randomization studies revealed that elevated plasma BCAAs portend higher BP in humans. Together, these data indicate that BCAA oxidation lowers vascular resistance, perhaps in part explaining cardioprotection in HF that is not mediated directly in cardiomyocytes.
Keywords: BCAA; Mendelian randomization; blood pressure; branched-chain amino acid; cardiac metabolism; cardiovascular metabolism; heart failure; hypertension; metabolomics.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests Z.A. received consulting fees from Pfizer. J.W.E. received consulting fees, unassociated with this work, from Jannsen Pharmaceuticals and Mitobridge, Inc. J.D.R. is an advisor and stockholder in Colorado Research Partners, Empress Therapeutics, L.E.A.F. Pharmaceuticals, Bantam Pharmaceuticals, Barer Institute, and Rafael Holdings; a founder, director, and stockholder of Farber Partners, Serien Therapeutics, and Sofro Pharmaceuticals; a founder and stockholder in Toran Therapeutics; inventor of patents held by Princeton University; and a director of the Princeton University-PKU Shenzhen collaboration.
Figures

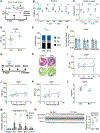

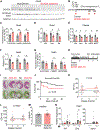
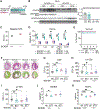


References
-
- Abel ED, Kaulbach HC, Tian R, Hopkins JCA, Duffy J, Doetschman T, Minnemann T, Boers M-E, Hadro E, Oberste-Berghaus C, et al. (1999). Cardiac hypertrophy with preserved contractile function after selective deletion of GLUT4 from the heart. J Clin Invest 104, 1703–1714. 10.1172/jci7605. - DOI - PMC - PubMed
-
- Bhattacharya S, Granger CB, Craig D, Haynes C, Bain J, Stevens RD, Hauser ER, Newgard CB, Kraus WE, Newby LK, et al. (2014). Validation of the association between a branched chain amino acid metabolite profile and extremes of coronary artery disease in patients referred for cardiac catheterization. Atherosclerosis 232, 191–196. 10.1016/j.atherosclerosis.2013.10.036. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- T32 CA257957/CA/NCI NIH HHS/United States
- S10 OD025098/OD/NIH HHS/United States
- T32 HL007954/HL/NHLBI NIH HHS/United States
- F30 HL142186/HL/NHLBI NIH HHS/United States
- R01 HL105993/HL/NHLBI NIH HHS/United States
- P30 DK019525/DK/NIDDK NIH HHS/United States
- R01 HL152446/HL/NHLBI NIH HHS/United States
- R01 DK114103/DK/NIDDK NIH HHS/United States
- F32 HL151146/HL/NHLBI NIH HHS/United States
- R01 HL149891/HL/NHLBI NIH HHS/United States
- R01 AG062140/AG/NIA NIH HHS/United States
- T32 HL007843/HL/NHLBI NIH HHS/United States
- R01 CA248315/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

