Mesenchymal stem cells exosomal let-7a-5p improve autophagic flux and alleviate liver injury in acute-on-chronic liver failure by promoting nuclear expression of TFEB
- PMID: 36224178
- PMCID: PMC9556718
- DOI: 10.1038/s41419-022-05303-9
Mesenchymal stem cells exosomal let-7a-5p improve autophagic flux and alleviate liver injury in acute-on-chronic liver failure by promoting nuclear expression of TFEB
Abstract
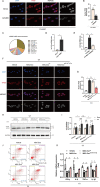
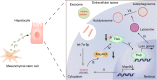
Acute-on-chronic liver failure is a distinct clinical syndrome characterized by a dysregulated immune response and extensive hepatocyte death without satisfactory therapies. As a cytoplasmic degradative and quality-control process, autophagy was implicated in maintaining intracellular homeostasis, and decreased hepatic autophagy was found in many liver diseases and contributes to disease pathogenesis. Previously, we identified the therapeutic potential of mesenchymal stem cells (MSCs) in ACLF patients; however, the intrinsic mechanisms are incompletely understood. Herein, we showed that MSCs restored the impaired autophagic flux and alleviated liver injuries in ACLF mice, but these effects were abolished when autophago-lysosomal maturation was inhibited by leupeptin (leu), suggesting that MSCs exerted their hepatoprotective function in a pro-autophagic dependent manner. Moreover, we described a connection between transcription factor EB (TFEB) and autophagic activity in this context, as evidenced by increased nuclei translocation of TFEB elicited by MSCs were capable of promoting liver autophagy. Mechanistically, we confirmed that let-7a-5p enriched in MSCs derived exosomes (MSC-Exo) could activate autophagy by targeting MAP4K3 to reduce TFEB phosphorylation, and MAP4K3 knockdown partially attenuates the effect of anti-let-7a-5p oligonucleotide via decreasing the inflammatory response, in addition, inducing autophagy. Altogether, these findings revealed that the hepatoprotective effect of MSCs may partially profit from its exosomal let-7a-5p mediating autophagy repairment, which may provide new insights for the therapeutic target of ACLF treatment.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Phillygenin ameliorates nonalcoholic fatty liver disease via TFEB-mediated lysosome biogenesis and lipophagy.Phytomedicine. 2022 Aug;103:154235. doi: 10.1016/j.phymed.2022.154235. Epub 2022 Jun 3. Phytomedicine. 2022. PMID: 35716542
-
Mesenchymal stem cell-regulated miRNA-mRNA landscape in acute-on-chronic liver failure.Genomics. 2023 Nov;115(6):110737. doi: 10.1016/j.ygeno.2023.110737. Epub 2023 Nov 4. Genomics. 2023. PMID: 37926353
-
Transplanted Mesenchymal Stem Cells Reduce Autophagic Flux in Infarcted Hearts via the Exosomal Transfer of miR-125b.Circ Res. 2018 Aug 17;123(5):564-578. doi: 10.1161/CIRCRESAHA.118.312758. Circ Res. 2018. PMID: 29921652
-
TFEB Biology and Agonists at a Glance.Cells. 2021 Feb 5;10(2):333. doi: 10.3390/cells10020333. Cells. 2021. PMID: 33562649 Free PMC article. Review.
-
TFEB in Alzheimer's disease: From molecular mechanisms to therapeutic implications.Neurobiol Dis. 2022 Oct 15;173:105855. doi: 10.1016/j.nbd.2022.105855. Epub 2022 Aug 27. Neurobiol Dis. 2022. PMID: 36031168 Review.
Cited by
-
Strategies to improve the therapeutic efficacy of mesenchymal stem cell-derived extracellular vesicle (MSC-EV): a promising cell-free therapy for liver disease.Front Bioeng Biotechnol. 2023 Dec 13;11:1322514. doi: 10.3389/fbioe.2023.1322514. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 38155924 Free PMC article. Review.
-
Role of mesenchymal stem cell-derived exosomes in the regeneration of different tissues.J Biol Eng. 2024 Jun 6;18(1):36. doi: 10.1186/s13036-024-00431-6. J Biol Eng. 2024. PMID: 38845032 Free PMC article. Review.
-
Mesenchymal stromal cells in hepatic fibrosis/cirrhosis: from pathogenesis to treatment.Cell Mol Immunol. 2023 Jun;20(6):583-599. doi: 10.1038/s41423-023-00983-5. Epub 2023 Feb 24. Cell Mol Immunol. 2023. PMID: 36823236 Free PMC article. Review.
-
Small extracellular vesicles from young adipose-derived stem cells ameliorate age-related changes in the heart of old mice.Stem Cell Res Ther. 2025 Mar 13;16(1):138. doi: 10.1186/s13287-025-04255-z. Stem Cell Res Ther. 2025. PMID: 40082997 Free PMC article.
-
Increased DNMT1 Involvement in the Activation of LO2 Cell Death Induced by Silver Nanoparticles via Promoting TFEB-Dependent Autophagy.Toxics. 2023 Sep 4;11(9):751. doi: 10.3390/toxics11090751. Toxics. 2023. PMID: 37755761 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

