Development and Evaluation of Dissolving Microarray Patches for Co-administered and Repeated Intradermal Delivery of Long-acting Rilpivirine and Cabotegravir Nanosuspensions for Paediatric HIV Antiretroviral Therapy
- PMID: 36224503
- PMCID: PMC10421828
- DOI: 10.1007/s11095-022-03408-6
Development and Evaluation of Dissolving Microarray Patches for Co-administered and Repeated Intradermal Delivery of Long-acting Rilpivirine and Cabotegravir Nanosuspensions for Paediatric HIV Antiretroviral Therapy
Abstract
Purpose: Whilst significant progress has been made to defeat HIV infection, the efficacy of antiretroviral (ARV) therapy in the paediatric population is often hindered by poor adherence. Currently, two long-acting (LA) intramuscular injectable nanosuspensions of rilpivirine (RPV) and cabotegravir (CAB) are in clinical development for paediatric populations. However, administration requires access to healthcare resources, is painful, and can result in needle-stick injuries to the end user. To overcome these barriers, this proof-of-concept study was developed to evaluate the intradermal delivery of RPV LA and CAB LA via self-disabling dissolving microarray patches (MAPs).
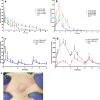
Methods: Dissolving MAPs of two conformations, a conventional pyramidal and a bilayer design, were formulated, with various nanosuspensions of RPV and CAB incorporated within the respective MAP matrix. MAPs were mechanically robust and were capable of penetrating ex vivo skin with intradermal ARV deposition.
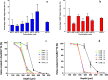
Results: In a single-dose in vivo study in rats, all ARV MAPs demonstrated sustained release profiles, with therapeutically relevant plasma concentrations of RPV and CAB detected to at least 63 and 28 d, respectively. In a multi-dose in vivo study, repeated MAP applications at 14-d intervals maintained therapeutically relevant plasma concentrations throughout the duration of the study.
Conclusions: These results illustrate the potential of the platform to repeatedly maintain plasma concentrations for RPV and CAB. As such, these MAPs could represent a viable option to improve adherence in the paediatric population, one that is capable of being painlessly administered in the comfort of the patient's own home on a biweekly or less frequent basis.
Keywords: AIDS, acquired immune deficiency syndrome; CAB, cabotegravir; HIV, human immunodeficiency virus; MAP, microarray patch; RPV, rilpivirine.
© 2022. The Author(s).
Conflict of interest statement
Ryan Donnelly is an inventor of patents that have been licenced to companies developing microarray patch-based products and is a paid advisor to a number of companies working towards commercialisation of this technology. The resulting potential conflict of interest has been disclosed and is managed by Queen’s University Belfast. The companies had no role in the design of the study; in the collection, analyses, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results. No other author has any competing interests.
Figures







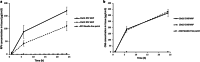
Similar articles
-
Intradermal delivery of the antiretroviral drugs cabotegravir and rilpivirine by dissolving microarray patches: Investigation of lymphatic uptake.J Control Release. 2024 Feb;366:548-566. doi: 10.1016/j.jconrel.2024.01.010. Epub 2024 Jan 13. J Control Release. 2024. PMID: 38211640
-
Patient-Reported Outcomes Through 1 Year of an HIV-1 Clinical Trial Evaluating Long-Acting Cabotegravir and Rilpivirine Administered Every 4 or 8 Weeks (ATLAS-2M).Patient. 2021 Nov;14(6):849-862. doi: 10.1007/s40271-021-00524-0. Epub 2021 May 31. Patient. 2021. PMID: 34056699 Free PMC article. Clinical Trial.
-
Design, formulation and evaluation of novel dissolving microarray patches containing a long-acting rilpivirine nanosuspension.J Control Release. 2018 Dec 28;292:119-129. doi: 10.1016/j.jconrel.2018.11.002. Epub 2018 Nov 2. J Control Release. 2018. PMID: 30395897 Free PMC article.
-
Practical dosing guidance for the management of clinician-administered injections of long-acting cabotegravir and rilpivirine.Ther Adv Infect Dis. 2023 Dec 13;10:20499361231214626. doi: 10.1177/20499361231214626. eCollection 2023 Jan-Dec. Ther Adv Infect Dis. 2023. PMID: 38107552 Free PMC article. Review.
-
Cabotegravir/Rilpivirine: the last FDA-approved drug to treat HIV.Expert Rev Anti Infect Ther. 2022 Aug;20(8):1135-1147. doi: 10.1080/14787210.2022.2081153. Epub 2022 Jun 13. Expert Rev Anti Infect Ther. 2022. PMID: 35596583 Review.
Cited by
-
Delivery of Islatravir via High Drug-Load, Long-acting Microarray Patches for the Prevention or Treatment of Human Immunodeficiency Virus.Adv Healthc Mater. 2025 Mar;14(7):e2403615. doi: 10.1002/adhm.202403615. Epub 2025 Jan 22. Adv Healthc Mater. 2025. PMID: 39840486 Free PMC article.
-
A Bilayer Microarray Patch (MAP) for HIV Pre-Exposure Prophylaxis: The Role of MAP Designs and Formulation Composition in Enhancing Long-Acting Drug Delivery.Pharmaceutics. 2024 Jan 20;16(1):142. doi: 10.3390/pharmaceutics16010142. Pharmaceutics. 2024. PMID: 38276512 Free PMC article.
-
Poly(acrylic acid)/Poly(vinyl alcohol) Microarray Patches for Continuous Transdermal Delivery of Levodopa and Carbidopa: In Vitro and In Vivo Studies.Pharmaceutics. 2024 May 17;16(5):676. doi: 10.3390/pharmaceutics16050676. Pharmaceutics. 2024. PMID: 38794339 Free PMC article.
-
Deferasirox Nanosuspension Loaded Dissolving Microneedles for Intradermal Delivery.Pharmaceutics. 2022 Dec 15;14(12):2817. doi: 10.3390/pharmaceutics14122817. Pharmaceutics. 2022. PMID: 36559310 Free PMC article.
-
The promise of microneedle technologies for drug delivery.Drug Deliv Transl Res. 2024 Mar;14(3):573-580. doi: 10.1007/s13346-023-01430-8. Epub 2023 Oct 2. Drug Deliv Transl Res. 2024. PMID: 37783973
References
-
- UNAIDS. Global HIV & AIDS statistics - Fact sheet (2021). https://www.unaids.org/en/resources/fact-sheet. Accessed 20 Jan 2022.
-
- Ciaranello AL, Perez F, Keatinge J, Park J-E, Engelsmann B, Maruva M, et al. What will it take to eliminate pediatric HIV? Reaching WHO target rates of mother-to-child HIV transmission in Zimbabwe: a model-based analysis. PLOS Med. 2012;9:1–15. doi: 10.1371/journal.pmed.1001156. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

