Antimalarial potential of compounds isolated from Mammea siamensis T. Anders. flowers: in vitro and molecular docking studies
- PMID: 36224571
- PMCID: PMC9554980
- DOI: 10.1186/s12906-022-03742-7
Antimalarial potential of compounds isolated from Mammea siamensis T. Anders. flowers: in vitro and molecular docking studies
Abstract
Background: The emergence of antimalarial drug resistance encourages the search for new antimalarial agents. Mammea siamensis belongs to the Calophyllaceae family, which is a medicinal plant that is used in traditional Thai preparations. The hexane and dichloromethane extracts of this plant were found to have potent antimalarial activity. Therefore, this study aimed to isolate active compounds from M. siamensis flowers and evaluate their antimalarial potential and their interactions with Plasmodium falciparum lactate dehydrogenase (PfLDH).
Methods: The compounds from M. siamensis flowers were isolated by chromatographic techniques and evaluated for their antimalarial activity against chloroquine (CQ)-resistant P. falciparum (K1) strains using a parasite lactate dehydrogenase (pLDH) assay. Interactions between the isolated compounds and the PfLDH enzyme were investigated using a molecular docking method.
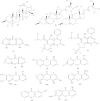
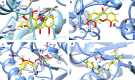
Results: The isolation produced the following thirteen compounds: two terpenoids, lupeol (1) and a mixture of β-sitosterol and stigmasterol (5); two mammea coumarins, mammea A/AA cyclo D (6) and mammea A/AA cyclo F (7); and nine xanthones, 4,5-dihydroxy-3-methoxyxanthone (2), 4-hydroxyxanthone (3), 1,7-dihydroxyxanthone (4), 1,6-dihydroxyxanthone (8), 1-hydroxy-5,6,7-trimethoxyxanthone (9), 3,4,5-trihydroxyxanthone (10), 5-hydroxy-1-methoxyxanthone (11), 2-hydroxyxanthone (12), and 1,5-dihydroxy-6-methoxyxanthone (13). Compound 9 exhibited the most potent antimalarial activity with an IC50 value of 9.57 µM, followed by 10, 1, 2 and 13 with IC50 values of 15.48, 18.78, 20.96 and 22.27 µM, respectively. The molecular docking results indicated that 9, which exhibited the most potent activity, also had the best binding affinity to the PfLDH enzyme in terms of its low binding energy (-7.35 kcal/mol) and formed interactions with ARG109, ASN140, and ARG171.
Conclusion: These findings revealed that isolated compounds from M. siamensis flowers exhibited antimalarial activity. The result suggests that 1-hydroxy-5,6,7-trimethoxyxanthone is a possible lead structure as a potent inhibitor of the PfLDH enzyme.
Keywords: Anti-malarial activity; Mammea siamensis; Molecular docking; Plasmodium falciparum; Xanthone.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests regarding the publication of this paper.
Figures
References
-
- WHO . World Malaria Report 2020. Geneva: World Health Organization; 2020.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

