Striatal miR-183-5p inhibits methamphetamine-induced locomotion by regulating glucocorticoid receptor signaling
- PMID: 36225577
- PMCID: PMC9549132
- DOI: 10.3389/fphar.2022.997701
Striatal miR-183-5p inhibits methamphetamine-induced locomotion by regulating glucocorticoid receptor signaling
Abstract
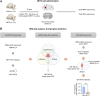
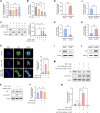
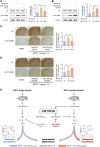
MicroRNA (miRNA)-mediated striatal gene regulation may play an important role in methamphetamine (METH) addiction. This study aimed to identify changes in novel miRNAs and their target genes during METH self-administration and investigate their roles in METH-induced locomotion. RNA sequencing analysis revealed that mir-183-5p was upregulated in the striatum of METH self-administered rats, and target gene prediction revealed that the glucocorticoid receptor (GR) gene, Nr3c1, was a potential target gene for mir-183-5p. We confirmed that single and repeated METH administrations increased METH-induced locomotion and plasma corticosterone levels in rats. Additionally, increased miR-185-5p expression and decreased GR gene expression were observed only in the repeated-METH-injection group but not in the single-injection group. We then investigated the effects of miR-183-5p on METH-induced locomotion using a miR-183-5p mimic and inhibitor. Injection of a mir-183-5p mimic in the striatum of rats attenuated METH-induced locomotion, whereas injection of a miR-183-5p inhibitor enhanced the locomotor activity in METH-administered rats. Furthermore, the miR-183-5p mimic reduced the phosphorylation of tyrosine hydroxylase (TH) whereas the inhibitor increased it. Taken together, these results indicate that repeated METH injections increase striatal miR-183-5p expression and regulate METH-induced locomotion by regulating GR expression in rats, thereby suggesting a potential role of miR-183-5p as a novel regulator of METH-induced locomotion.
Keywords: RNA sequencing; glucocorticoid receptor; locomotor activity; methamphetamine; microRNA; self-administration.
Copyright © 2022 Song, Jang, Jang, Kim, Kim, Son, Choi, Lee and Jeong.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
From dysregulated microRNAs to structural alterations in the striatal region of METH-injected rats.J Chem Neuroanat. 2020 Nov;109:101854. doi: 10.1016/j.jchemneu.2020.101854. Epub 2020 Aug 11. J Chem Neuroanat. 2020. PMID: 32795519
-
Rhynchophylline inhibits methamphetamine dependence via modulating the miR-181a-5p/GABRA1 axis.J Ethnopharmacol. 2023 Oct 5;314:116635. doi: 10.1016/j.jep.2023.116635. Epub 2023 May 13. J Ethnopharmacol. 2023. PMID: 37182675
-
MicroRNA expression signature of methamphetamine use and addiction in the rat nucleus accumbens.Metab Brain Dis. 2017 Dec;32(6):1767-1783. doi: 10.1007/s11011-017-0061-x. Epub 2017 Jul 5. Metab Brain Dis. 2017. PMID: 28681200
-
Regulation of microRNA-29c in the nucleus accumbens modulates methamphetamine -induced locomotor sensitization in mice.Neuropharmacology. 2019 Apr;148:160-168. doi: 10.1016/j.neuropharm.2019.01.007. Epub 2019 Jan 9. Neuropharmacology. 2019. PMID: 30639389
-
Locomotion changes in methamphetamine and amphetamine withdrawal: a systematic review.Front Pharmacol. 2024 Jul 17;15:1428492. doi: 10.3389/fphar.2024.1428492. eCollection 2024. Front Pharmacol. 2024. PMID: 39086393 Free PMC article.
Cited by
-
Identification of Potential Biomarkers for Diagnosis of Patients with Methamphetamine Use Disorder.Int J Mol Sci. 2023 May 12;24(10):8672. doi: 10.3390/ijms24108672. Int J Mol Sci. 2023. PMID: 37240016 Free PMC article.
-
Uncovering transcriptomic biomarkers for enhanced diagnosis of methamphetamine use disorder: a comprehensive review.Front Psychiatry. 2024 Jan 8;14:1302994. doi: 10.3389/fpsyt.2023.1302994. eCollection 2023. Front Psychiatry. 2024. PMID: 38260797 Free PMC article.
-
Identification of the circRNA-miRNA-mRNA network for treating methamphetamine-induced relapse and behavioral sensitization with cannabidiol.CNS Neurosci Ther. 2024 May;30(5):e14737. doi: 10.1111/cns.14737. CNS Neurosci Ther. 2024. PMID: 38702929 Free PMC article.
-
Brain microRNA profiles after exposure to heroin in rats.Exp Brain Res. 2024 Dec 13;243(1):24. doi: 10.1007/s00221-024-06972-y. Exp Brain Res. 2024. PMID: 39671092
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

