Wood degradation by Fomitiporia mediterranea M. Fischer: Physiologic, metabolomic and proteomic approaches
- PMID: 36226293
- PMCID: PMC9549746
- DOI: 10.3389/fpls.2022.988709
Wood degradation by Fomitiporia mediterranea M. Fischer: Physiologic, metabolomic and proteomic approaches
Abstract
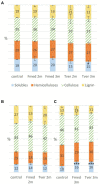
Fomitiporia mediterranea (Fmed) is one of the main fungal species found in grapevine wood rot, also called "amadou," one of the most typical symptoms of grapevine trunk disease Esca. This fungus is functionally classified as a white-rot, able to degrade all wood structure polymers, i.e., hemicelluloses, cellulose, and the most recalcitrant component, lignin. Specific enzymes are secreted by the fungus to degrade those components, namely carbohydrate active enzymes for hemicelluloses and cellulose, which can be highly specific for given polysaccharide, and peroxidases, which enable white-rot to degrade lignin, with specificities relating to lignin composition as well. Furthermore, besides polymers, a highly diverse set of metabolites often associated with antifungal activities is found in wood, this set differing among the various wood species. Wood decayers possess the ability to detoxify these specific extractives and this ability could reflect the adaptation of these fungi to their specific environment. The aim of this study is to better understand the molecular mechanisms used by Fmed to degrade wood structure, and in particular its potential adaptation to grapevine wood. To do so, Fmed was cultivated on sawdust from different origins: grapevine, beech, and spruce. Carbon mineralization rate, mass loss, wood structure polymers contents, targeted metabolites (extractives) and secreted proteins were measured. We used the well-known white-rot model Trametes versicolor for comparison. Whereas no significant degradation was observed with spruce, a higher mass loss was measured on Fmed grapevine culture compared to beech culture. Moreover, on both substrates, a simultaneous degradation pattern was demonstrated, and proteomic analysis identified a relative overproduction of oxidoreductases involved in lignin and extractive degradation on grapevine cultures, and only few differences in carbohydrate active enzymes. These results could explain at least partially the adaptation of Fmed to grapevine wood structural composition compared to other wood species, and suggest that other biotic and abiotic factors should be considered to fully understand the potential adaptation of Fmed to its ecological niche. Proteomics data are available via ProteomeXchange with identifier PXD036889.
Keywords: Esca; Fomitiporia mediterranea; adaptation; grapevine wood; white rot.
Copyright © 2022 Schilling, Maia-Grondard, Baltenweck, Robert, Hugueney, Bertsch, Farine and Gelhaye.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures






References
-
- Abou-Mansour E., Polier J., Pezet R., Tabacchi R. (2009). Purification and partial characterisation of a 60 KDa laccase from. Phytopathol. Mediterr. 48, 447–453.
-
- AFMB, Architecture et Fonction des Macromolécules Biologiques UMR7257- CNRS - Université d’Aix-Marseille (2022a). Carbohydrate Esterase Family Classification. CAZy.org. Available at: http://www.cazy.org/Carbohydrate-Esterases.html (Accessed May 7, 2022).
-
- AFMB, Architecture et Fonction des Macromolécules Biologiques UMR7257- CNRS - Université d’Aix-Marseille (2022b). Glycoside Hydrolase Family Classification. CAZy.org. Available at: http://www.cazy.org/Glycoside-Hydrolases.html (Accessed May 7, 2022).
LinkOut - more resources
Full Text Sources

