CXCR1 participates in bone cancer pain induced by Walker 256 breast cancer cells in female rats
- PMID: 36227008
- PMCID: PMC9638694
- DOI: 10.1177/17448069221135743
CXCR1 participates in bone cancer pain induced by Walker 256 breast cancer cells in female rats
Abstract
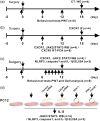
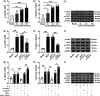
Bone cancer pain (BCP) is a clinically intractable mixed pain, involving inflammation and neuropathic pain, and its mechanisms remain unclear. CXC chemokine receptor 1 (CXCR1, IL-8RA) and 2 (CXCR2, IL-8RB) are high-affinity receptors for interleukin 8 (IL8). According to previous studies, CXCR2 plays a crucial role in BCP between astrocytes and neurons, while the role of CXCR1 remains unclear. The objective of this study was to investigate the role of CXCR1 in BCP. We found that CXCR1 expression increased in the spinal dorsal horn. Intrathecal injection of CXCR1 siRNA effectively attenuated mechanical allodynia and pain-related behaviors in rats. It was found that CXCR1 was predominantly co-localized with neurons. Intrathecal injection of CXCR1-siRNA reduced phosphorylated JAK2/STAT3 protein levels and the NLRP3 inflammasome (NLRP3, caspase1, and IL-1β) levels. Furthermore, in vitro cytological experiments confirmed this conclusion. The study results suggest that the spinal chemokine receptor CXCR1 activation mediates BCP through JAK2/STAT3 signaling pathway and NLRP3 inflammasome (NLRP3, caspase1, and IL-1β).
Keywords: CXCR1; JAK2/STAT3 signaling pathway; NLRP3 inflammasome; bone cancer pain; spinal cord.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

