Adjuvant Capecitabine Following Concurrent Chemoradiotherapy in Locoregionally Advanced Nasopharyngeal Carcinoma: A Randomized Clinical Trial
- PMID: 36227615
- PMCID: PMC9562101
- DOI: 10.1001/jamaoncol.2022.4656
Adjuvant Capecitabine Following Concurrent Chemoradiotherapy in Locoregionally Advanced Nasopharyngeal Carcinoma: A Randomized Clinical Trial
Abstract
Importance: Induction or adjuvant chemotherapy with concurrent chemoradiotherapy (CCRT) are first-line treatment options for treatment of locoregionally advanced nasopharyngeal carcinoma (LA-NPC). Adjuvant platinum regimens are, however, poorly tolerated, highlighting the unmet need for an efficacious, tolerable adjuvant regimen.
Objective: To investigate the efficacy and safety of adjuvant capecitabine with CCRT for the treatment of patients with LA-NPC.
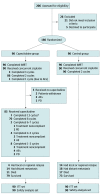
Design, setting, and participants: This open-label randomized clinical trial recruited patients from March 31, 2014, to July 27, 2018, at 3 institutions in China, with at least 3 years of follow-up. The data collection cutoff date was February 9, 2022. Eligibility included stage III-IVb nasopharyngeal carcinoma and at least 1 of the following: T3-4N2 or T1-4N3; plasma Epstein-Barr virus DNA titer higher than 20 000 copies/mL; primary gross tumor volume larger than 30.0 cm3; fluorodeoxyglucose F 18 positron emission tomography/computed tomography maximum standard uptake value of the primary gross tumor volume larger than 10.0; or multiple nodal metastases and any larger than 4.0 cm.
Interventions: Patients were randomly assigned 1:1 to receive either capecitabine (1000 mg/m2 twice daily for 14 days every 3 weeks for 8 cycles) or observation following CCRT (100 mg/m2 cisplatin every 3 weeks for 2 to 3 cycles, depending on duration of radiotherapy).
Main outcomes and measures: Failure-free survival in the intention-to-treat cohort was assessed using Kaplan-Meier survival curves compared with the log-rank test. Unstratified Cox proportional hazards regression models were used to estimate hazard ratios, with corresponding 95% CIs based on the Wald test.
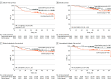
Results: There were 180 patients enrolled (median [IQR] age, 47 [40-55] years; 143 [79.4%] men). Among 90 patients in the capecitabine group, 76 (84.4%) had at least 2 high-risk factors; among 90 patients in the control group, 80 (88.9%) had at least 2 high-risk factors. All patients completed CCRT, except 1 patient in the capecitabine group who received 1 cycle of cisplatin. Of the 90 patients in the capecitabine group, 85 (94.4%) received capecitabine, with 71 (78.9%) completing 8 cycles. With a median (IQR) follow-up of 58.0 (49.5-80.1) months, 18 events were recorded in the capecitabine group vs 31 events in the control group. Failure-free survival was improved with adjuvant capecitabine (3 years, 83.3% vs 72.2%; 5 years, 78.5% vs 65.9%; hazard ratio, 0.53 [95% CI, 0.30-0.94]; P = .03). The incidence of grade 3 treatment-related adverse events (TRAEs) was higher in the capecitabine group than in the control group (54 of 90 patients [60.0%] vs 46 of 90 patients [51.1%]). Treatment-related adverse events included xerostomia (17 [18.9%] vs 9 [10.0%] patients), mucositis (21 [23.3%] vs 15 [16.7%] patients), and anorexia (8 [8.9%] vs 4 [4.4%] patients). The incidence of grade 3 delayed treatment-related adverse events was comparable in both groups (9 of 83 [10.8%] vs 7 of 81 [8.6%] patients).
Conclusions and relevance: In this randomized clinical trial, adjuvant capecitabine at the full dose following CCRT was well tolerated and improved failure-free survival among patients with LA-NPC and high-risk factors. Further investigations assessing optimal dose and duration are warranted.
Trial registration: ClinicalTrials.gov Identifier: NCT02143388.
Conflict of interest statement
Figures
References
Associated data
LinkOut - more resources
Full Text Sources
Medical

