Spatial clustering in vaccination hesitancy: The role of social influence and social selection
- PMID: 36227809
- PMCID: PMC9562150
- DOI: 10.1371/journal.pcbi.1010437
Spatial clustering in vaccination hesitancy: The role of social influence and social selection
Abstract
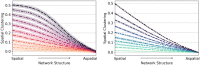
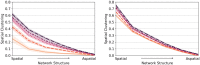
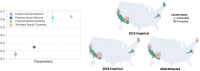
The phenomenon of vaccine hesitancy behavior has gained ground over the last three decades, jeopardizing the maintenance of herd immunity. This behavior tends to cluster spatially, creating pockets of unprotected sub-populations that can be hotspots for outbreak emergence. What remains less understood are the social mechanisms that can give rise to spatial clustering in vaccination behavior, particularly at the landscape scale. We focus on the presence of spatial clustering, and aim to mechanistically understand how different social processes can give rise to this phenomenon. In particular, we propose two hypotheses to explain the presence of spatial clustering: (i) social selection, in which vaccine-hesitant individuals share socio-demographic traits, and clustering of these traits generates spatial clustering in vaccine hesitancy; and (ii) social influence, in which hesitant behavior is contagious and spreads through neighboring societies, leading to hesitant clusters. Adopting a theoretical spatial network approach, we explore the role of these two processes in generating patterns of spatial clustering in vaccination behaviors under a range of spatial structures. We find that both processes are independently capable of generating spatial clustering, and the more spatially structured the social dynamics in a society are, the higher spatial clustering in vaccine-hesitant behavior it realizes. Together, we demonstrate that these processes result in unique spatial configurations of hesitant clusters, and we validate our models of both processes with fine-grain empirical data on vaccine hesitancy, social determinants, and social connectivity in the US. Finally, we propose, and evaluate the effectiveness of two novel intervention strategies to diminish hesitant behavior. Our generative modeling approach informed by unique empirical data provides insights on the role of complex social processes in driving spatial heterogeneity in vaccine hesitancy.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Thangaraju Pugazhenthan and Venkatesan Sajitha. Who ten threats to global health in 2019: Antimicrobial resistance. Cukurova Medical Journal, 44(3):1150–1151, 2019.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

