The Hippo pathway links adipocyte plasticity to adipose tissue fibrosis
- PMID: 36229481
- PMCID: PMC9562301
- DOI: 10.1038/s41467-022-33800-0
The Hippo pathway links adipocyte plasticity to adipose tissue fibrosis
Abstract
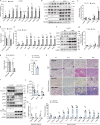
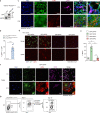
Fibrosis disrupts adipose tissue (AT) homeostasis and exacerbates metabolic dysfunction upon chronic caloric excess. The molecular mechanisms linking adipocyte plasticity to AT fibrosis are largely unknown. Here we show that the Hippo pathway is coupled with TGFβ signaling to orchestrate a cellular and/or functional shift of adipocytes from energy storage to extracellular matrix (ECM) remodeling in AT fibrosis. We found that Lats1/2-knockout adipocytes could dedifferentiate into DPP4+ progenitor cells and convert to DPP4- myofibroblasts upon TGFβ stimulation. On the other hand, Hippo pathway inhibition during obesity impaired adipocyte identity while promoted ECM remodeling activity of adipocytes. Macrophages recruited by CCL2 produced TGFβ to accelerate AT fibrosis. YAP and TAZ, the Hippo downstream effectors, enhanced SMAD2 stability to promote fibrotic responses. Importantly, inhibition of YAP/TAZ activity in obese mice markedly relieved AT fibrosis and improved metabolic homeostasis. Together, our findings identify the Hippo pathway as a molecular switch in the initiation and development of AT fibrosis, implying it as a therapeutic target.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

