A potent antibiotic-loaded bone-cement implant against staphylococcal bone infections
- PMID: 36229662
- PMCID: PMC10101771
- DOI: 10.1038/s41551-022-00950-x
A potent antibiotic-loaded bone-cement implant against staphylococcal bone infections
Abstract
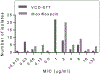
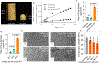
New antibiotics should ideally exhibit activity against drug-resistant bacteria, delay the development of bacterial resistance to them and be suitable for local delivery at desired sites of infection. Here, we report the rational design, via molecular-docking simulations, of a library of 17 candidate antibiotics against bone infection by wild-type and mutated bacterial targets. We screened this library for activity against multidrug-resistant clinical isolates and identified an antibiotic that exhibits potent activity against resistant strains and the formation of biofilms, decreases the chances of bacterial resistance and is compatible with local delivery via a bone-cement matrix. The antibiotic-loaded bone cement exhibited greater efficacy than currently used antibiotic-loaded bone cements against staphylococcal bone infections in rats. Potent and locally delivered antibiotic-eluting polymers may help address antimicrobial resistance.
© 2022. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Competing interests
Sumana Ghosh, M.S., R.S., S. Sadhasivam, A.N., A.B., S Saini, N.T., H.S., S. Gupta, A.C., M.K.G., and Shamik Ghosh are employees of Vyome Therapeutics Limited. Sumana Ghosh and Shamik Ghosh hold equity in Vyome Therapeutics Inc. S. Sengupta is a co-founder and board member of Vyome Therapeutics Limited, and owns equity in Vyome Therapeutics Inc. H.L.J. is a founder of Curer Inc. and holds equity in the company. S.S.V. and Sudip Ghosh declare no competing interests.
Figures





References
-
- Morris K Battle against antibiotic resistance is being lost. The Lancet Infectious Diseases 7, 509, doi:10.1016/S1473-3099(07)70173-1 (2007). - DOI
-
- Teillant A, Gandra S, Barter D, Morgan DJ & Laxminarayan R Potential burden of antibiotic resistance on surgery and cancer chemotherapy antibiotic prophylaxis in the USA: a literature review and modelling study. Lancet Infect Dis 15, 1429–1437, doi:10.1016/s1473-3099(15)00270-4 (2015). - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

