Ultrasound guided corticosteroids sacroiliac joint injections (SIJIs) in the management of active sacroiliitis: a real-life prospective experience
- PMID: 36229757
- PMCID: PMC10247913
- DOI: 10.1007/s40477-022-00736-6
Ultrasound guided corticosteroids sacroiliac joint injections (SIJIs) in the management of active sacroiliitis: a real-life prospective experience
Abstract
Introduction: Active sacroiliitis represents the hallmark of axial spondyloarthritis (axSpA) and manifests as inflammatory low back pain associated with morning stiffness (MS). Sometimes, the combination of non-steroidal anti-inflammatory drugs (NSAIDs) and biological disease modifying drugs (bDMARDs) proves unsatisfactory in achieving a remission.
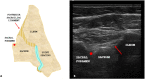
Materials and methods: We enrolled patients affected with active sacroiliitis confirmed via magnetic resonance imaging (MRI) and treated with a corticosteroid sacroiliac joint injection (SIJI) via ultrasound guidance. After SIJI, we evaluated visual-analogue scale (VAS) and MS pain changes. As controls, we selected axSpA patients starting bDMARDs.
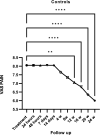
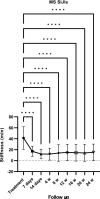
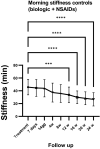
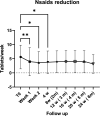
Results: We enrolled 26 patients (mean age 55 ± 14 years; 25 females and 1 male; > 95% treated with NSAIDs; 46% on bDMARDs; 75.82 ± 123 months) and examined a total of 47 treated joints. We detected a 48% reduction in VAS pain after 24 h. Moreover, we observed a significant reduction (p < 0.0001) of VAS pain between the baseline and every subsequent follow-up visit. Further, a significant difference in VAS pain compared to the baseline in the controls was observed starting from week 12. There was a significant reduction in MS after 1 week due to SIJIs, while in the controls the first significant change from the baseline in MS was detected after 12 weeks. The efficacy of infiltrative therapy lasted up to 6 months: persistent VAS as well as MS pain reduction was observed.
Conclusions: US-guided SIJI represents an effective and safe technique for patients who have active sacroiliitis yet are ineligible for biologic treatment or who experience unsatisfactory disease control despite receiving therapy.
Keywords: Active sacroiliitis; AxSpa; Morning stiffness; Pain; Sacroiliac joint injections; Ultrasound.
© 2022. Società Italiana di Ultrasonologia in Medicina e Biologia (SIUMB).
Conflict of interest statement
The authors deny any conflict of interest.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

