Preservation of the Hypoxic Transcriptome in Glioblastoma Patient-Derived Cell Lines Maintained at Lowered Oxygen Tension
- PMID: 36230775
- PMCID: PMC9564145
- DOI: 10.3390/cancers14194852
Preservation of the Hypoxic Transcriptome in Glioblastoma Patient-Derived Cell Lines Maintained at Lowered Oxygen Tension
Abstract
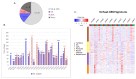
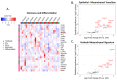
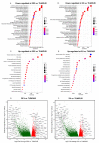
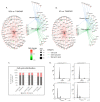
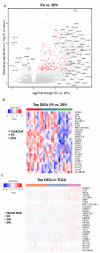
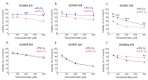
Despite numerous efforts aiming to characterise glioblastoma pathology (GBM) and discover new therapeutic strategies, GBM remains one of the most challenging tumours to treat. Here we propose the optimisation of in vitro culturing of GBM patient-derived cells, namely the establishment of GBM-derived cultures and their maintenance at oxygen tension mimicking oxygenation conditions occurring within the tumour. To globally analyse cell states, we performed the transcriptome analysis of GBM patient-derived cells kept as spheroids in serum-free conditions at the reduced oxygen tension (5% O2), cells cultured at atmospheric oxygen (20% O2), and parental tumour. Immune cells present in the tumour were depleted, resulting in the decreased expression of the immune system and inflammation-related genes. The expression of genes promoting cell proliferation and DNA repair was higher in GBM cell cultures when compared to the relevant tumour sample. However, lowering oxygen tension to 5% did not affect the proliferation rate and expression of cell cycle and DNA repair genes in GBM cell cultures. Culturing GBM cells at 5% oxygen was sufficient to increase the expression of specific stemness markers, particularly the PROM1 gene, without affecting neural cell differentiation markers. GBM spheroids cultured at 5% oxygen expressed higher levels of hypoxia-inducible genes, including those encoding glycolytic enzymes and pro-angiogenic factors. The genes up-regulated in cells cultured at 5% oxygen had higher expression in parental GBMs compared to that observed in 20% cell cultures, suggesting the preservation of the hypoxic component of GBM transcriptome at 5% oxygen and its loss in standard culture conditions. Evaluation of expression of those genes in The Cancer Genome Atlas dataset comprising samples of normal brain tissue, lower-grade gliomas and GBMs indicated the expression pattern of the indicated genes was specific for GBM. Moreover, GBM cells cultured at 5% oxygen were more resistant to temozolomide, the chemotherapeutic used in GBM therapy. The presented comparison of GBM cultures maintained at high and low oxygen tension together with analysis of tumour transcriptome indicates that lowering oxygen tension during cell culture may more allegedly reproduce tumour cell behaviour within GBM than standard culture conditions (e.g., atmospheric oxygen tension). Low oxygen culture conditions should be considered as a more appropriate model for further studies on glioblastoma pathology and therapy.
Keywords: 5% oxygen; DNA repair; RNA sequencing; cell cycle; glioblastoma; glioblastoma stem cells; hypoxia; inflammation; spheroid; temozolomide.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Wen P.Y., Weller M., Lee E.Q., Alexander B.M., Barnholtz-Sloan J.S., Barthel F.P., Batchelor T.T., Bindra R.S., Chang S.M., Chiocca E.A., et al. Glioblastoma in Adults: A Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) Consensus Review on Current Management and Future Directions. Neuro-Oncol. 2020;22:1073–1113. doi: 10.1093/neuonc/noaa106. - DOI - PMC - PubMed
-
- Patel A.P., Tirosh I., Trombetta J.J., Shalek A.K., Gillespie S.M., Wakimoto H., Cahill D.P., Nahed B.V., Curry W.T., Martuza R.L., et al. Single-Cell RNA-Seq Highlights Intratumoral Heterogeneity in Primary Glioblastoma. Science. 2014;344:1396–1401. doi: 10.1126/science.1254257. - DOI - PMC - PubMed
-
- Sottoriva A., Spiteri I., Piccirillo S.G.M., Touloumis A., Collins V.P., Marioni J.C., Curtis C., Watts C., Tavare S. Intratumor Heterogeneity in Human Glioblastoma Reflects Cancer Evolutionary Dynamics. Proc. Natl. Acad. Sci. USA. 2013;110:4009–4014. doi: 10.1073/pnas.1219747110. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

