Lung Subregion Partitioning by Incremental Dose Intervals Improves Omics-Based Prediction for Acute Radiation Pneumonitis in Non-Small-Cell Lung Cancer Patients
- PMID: 36230812
- PMCID: PMC9564373
- DOI: 10.3390/cancers14194889
Lung Subregion Partitioning by Incremental Dose Intervals Improves Omics-Based Prediction for Acute Radiation Pneumonitis in Non-Small-Cell Lung Cancer Patients
Abstract
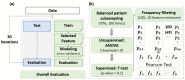
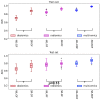
Purpose: To evaluate the effectiveness of features obtained from our proposed incremental-dose-interval-based lung subregion segmentation (IDLSS) for predicting grade ≥ 2 acute radiation pneumonitis (ARP) in lung cancer patients upon intensity-modulated radiotherapy (IMRT). (1) Materials and Methods: A total of 126 non-small-cell lung cancer patients treated with IMRT were retrospectively analyzed. Five lung subregions (SRs) were generated by the intersection of the whole lung (WL) and five sub-regions receiving incremental dose intervals. A total of 4610 radiomics features (RF) from pre-treatment planning computed tomographic (CT) and 213 dosiomics features (DF) were extracted. Six feature groups, including WL-RF, WL-DF, SR-RF, SR-DF, and the combined feature sets of WL-RDF and SR-RDF, were generated. Features were selected by using a variance threshold, followed by a Student t-test. Pearson’s correlation test was applied to remove redundant features. Subsequently, Ridge regression was adopted to develop six models for ARP using the six feature groups. Thirty iterations of resampling were implemented to assess overall model performance by using the area under the Receiver-Operating-Characteristic curve (AUC), accuracy, precision, recall, and F1-score. (2) Results: The SR-RDF model achieved the best classification performance and provided significantly better predictability than the WL-RDF model in training cohort (Average AUC: 0.98 ± 0.01 vs. 0.90 ± 0.02, p < 0.001) and testing cohort (Average AUC: 0.88 ± 0.05 vs. 0.80 ± 0.04, p < 0.001). Similarly, predictability of the SR-DF model was significantly stronger than that of the WL-DF model in training cohort (Average AUC: 0.88 ± 0.03 vs. 0.70 ± 0.030, p < 0.001) and in testing cohort (Average AUC: 0.74 ± 0.08 vs. 0.65 ± 0.06, p < 0.001). By contrast, the SR-RF model significantly outperformed the WL-RF model only in the training set (Average AUC: 0.93 ± 0.02 vs. 0.85 ± 0.03, p < 0.001), but not in the testing set (Average AUC: 0.79 ± 0.05 vs. 0.77 ± 0.07, p = 0.13). (3) Conclusions: Our results demonstrated that the IDLSS method improved model performance for classifying ARP with grade ≥ 2 when using dosiomics or combined radiomics-dosiomics features.
Keywords: dosiomics; multi-omics; radiation pneumonitis; radiomics; radiotherapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Multi-omics to predict acute radiation esophagitis in patients with lung cancer treated with intensity-modulated radiation therapy.Eur J Med Res. 2023 Mar 19;28(1):126. doi: 10.1186/s40001-023-01041-6. Eur J Med Res. 2023. PMID: 36935504 Free PMC article.
-
Function-Wise Dual-Omics analysis for radiation pneumonitis prediction in lung cancer patients.Front Pharmacol. 2022 Sep 19;13:971849. doi: 10.3389/fphar.2022.971849. eCollection 2022. Front Pharmacol. 2022. PMID: 36199694 Free PMC article.
-
Multi-institutional dose-segmented dosiomic analysis for predicting radiation pneumonitis after lung stereotactic body radiation therapy.Med Phys. 2021 Apr;48(4):1781-1791. doi: 10.1002/mp.14769. Epub 2021 Mar 2. Med Phys. 2021. PMID: 33576510
-
Dosiomics and radiomics-based prediction of pneumonitis after radiotherapy and immune checkpoint inhibition: The relevance of fractionation.Lung Cancer. 2024 Mar;189:107507. doi: 10.1016/j.lungcan.2024.107507. Epub 2024 Feb 17. Lung Cancer. 2024. PMID: 38394745
-
Radiation pneumonitis prediction after stereotactic body radiation therapy based on 3D dose distribution: dosiomics and/or deep learning-based radiomics features.Radiat Oncol. 2022 Nov 17;17(1):188. doi: 10.1186/s13014-022-02154-8. Radiat Oncol. 2022. PMID: 36397060 Free PMC article. Clinical Trial.
Cited by
-
Association of Multi-Phasic MR-Based Radiomic and Dosimetric Features with Treatment Response in Unresectable Hepatocellular Carcinoma Patients following Novel Sequential TACE-SBRT-Immunotherapy.Cancers (Basel). 2023 Feb 9;15(4):1105. doi: 10.3390/cancers15041105. Cancers (Basel). 2023. PMID: 36831445 Free PMC article.
-
Development and validation of a lung biological equivalent dose-based multiregional radiomic model for predicting symptomatic radiation pneumonitis after SBRT in lung cancer patients.Front Oncol. 2024 Dec 6;14:1489217. doi: 10.3389/fonc.2024.1489217. eCollection 2024. Front Oncol. 2024. PMID: 39711952 Free PMC article.
-
Multi-omics and Multi-VOIs to predict esophageal fistula in esophageal cancer patients treated with radiotherapy.J Cancer Res Clin Oncol. 2024 Jan 27;150(2):39. doi: 10.1007/s00432-023-05520-5. J Cancer Res Clin Oncol. 2024. PMID: 38280037 Free PMC article.
-
Multi-omics to predict acute radiation esophagitis in patients with lung cancer treated with intensity-modulated radiation therapy.Eur J Med Res. 2023 Mar 19;28(1):126. doi: 10.1186/s40001-023-01041-6. Eur J Med Res. 2023. PMID: 36935504 Free PMC article.
-
Multimodal Data Integration to Predict Severe Acute Oral Mucositis of Nasopharyngeal Carcinoma Patients Following Radiation Therapy.Cancers (Basel). 2023 Mar 29;15(7):2032. doi: 10.3390/cancers15072032. Cancers (Basel). 2023. PMID: 37046693 Free PMC article.
References
-
- Baisden J.M., Romney D.A., Reish A.G., Cai J., Sheng K., Jones D.R., Benedict S.H., Read P.W., Larner J.M. Dose as a function of lung volume and planned treatment volume in helical tomotherapy intensity-modulated radiation therapy-based stereotactic body radiation therapy for small lung tumors. Int. J. Radiat. Oncol. Biol. Phys. 2007;68:1229–1237. doi: 10.1016/j.ijrobp.2007.03.024. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

