Thyroid Hormone Induces Oral Cancer Growth via the PD-L1-Dependent Signaling Pathway
- PMID: 36231010
- PMCID: PMC9563246
- DOI: 10.3390/cells11193050
Thyroid Hormone Induces Oral Cancer Growth via the PD-L1-Dependent Signaling Pathway
Abstract
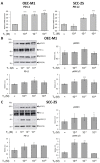
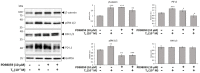
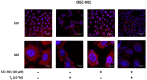
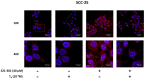
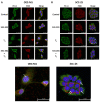
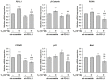
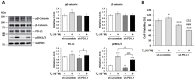
Oral cancer is a fatal disease, and its incidence in Taiwan is increasing. Thyroid hormone as L-thyroxine (T4) stimulates cancer cell proliferation via a receptor on integrin αvβ3 of plasma membranes. It also induces the expression of programmed death-ligand 1 (PD-L1) and cell proliferation in cancer cells. Thyroid hormone also activates β-catenin-dependent cell proliferation in cancer cells. However, the relationship between PD-L1 and cancer proliferation is not fully understood. In the current study, we investigated the role of inducible thyroid hormone-induced PD-L1-regulated gene expression and proliferation in oral cancer cells. Thyroxine bound to integrin αvβ3 to induce PD-L1 expressions via activation of ERK1/2 and signal transducer and activator of transcription 3 (STAT3). Inactivated STAT3 inhibited PD-L1 expression and nuclear PD-L1 accumulation. Inhibition of PD-L1 expression reduced β-catenin accumulation. Furthermore, nuclear PD-L1 formed a complex with nuclear proteins such as p300. Suppression PD-L1 expression by shRNA blocked not only expression of PD-L1 and β-catenin but also signal transduction, proliferative gene expressions, and cancer cell growth. In summary, thyroxine via integrin αvβ3 activated ERK1/2 and STAT3 to stimulate the PD-L1-dependent and β-catenin-related growth in oral cancer cells.
Keywords: PD-L1; oral cancer; thyroxine; β-catenin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Thyroid hormone-induced expression of inflammatory cytokines interfere with resveratrol-induced anti-proliferation of oral cancer cells.Food Chem Toxicol. 2019 Oct;132:110693. doi: 10.1016/j.fct.2019.110693. Epub 2019 Jul 20. Food Chem Toxicol. 2019. PMID: 31336132
-
Thyroxine inhibits resveratrol-caused apoptosis by PD-L1 in ovarian cancer cells.Endocr Relat Cancer. 2018 May;25(5):533-545. doi: 10.1530/ERC-17-0376. Epub 2018 Mar 19. Endocr Relat Cancer. 2018. PMID: 29555649
-
Actions of l-thyroxine and Nano-diamino-tetrac (Nanotetrac) on PD-L1 in cancer cells.Steroids. 2016 Oct;114:59-67. doi: 10.1016/j.steroids.2016.05.006. Epub 2016 May 21. Steroids. 2016. PMID: 27221508
-
Actions of Thyroid Hormones on Thyroid Cancers.Front Endocrinol (Lausanne). 2021 Jun 21;12:691736. doi: 10.3389/fendo.2021.691736. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34234745 Free PMC article. Review.
-
Regulation of PD-L1: a novel role of pro-survival signalling in cancer.Ann Oncol. 2016 Mar;27(3):409-16. doi: 10.1093/annonc/mdv615. Epub 2015 Dec 17. Ann Oncol. 2016. PMID: 26681673 Review.
Cited by
-
Mitogen-Activated Protein Kinase and Nuclear Hormone Receptor Crosstalk in Cancer Immunotherapy.Int J Mol Sci. 2023 Sep 4;24(17):13661. doi: 10.3390/ijms241713661. Int J Mol Sci. 2023. PMID: 37686465 Free PMC article. Review.
-
Thyroid disorders as predictors of cemiplimab efficacy in recurrent/metastatic cervical cancer: real-world evidence from Poland.Front Immunol. 2025 Jun 20;16:1604826. doi: 10.3389/fimmu.2025.1604826. eCollection 2025. Front Immunol. 2025. PMID: 40621463 Free PMC article.
-
Exploring Natural Compounds Targeting PD-L1 and STAT3: Toxicogenomic Analysis, Virtual Screening, Molecular Docking, ADMET Evaluation, and Biological Activity Prediction.Curr Comput Aided Drug Des. 2025;21(3):348-361. doi: 10.2174/0115734099307259240522093710. Curr Comput Aided Drug Des. 2025. PMID: 38808724
References
-
- Kao S.Y., Lim E. An overview of detection and screening of oral cancer in Taiwan. Chin. J. Dent. Res. 2015;18:7–12. - PubMed
-
- Hou J., Zhao R., Xia W., Chang C.W., You Y., Hsu J.M., Nie L., Chen Y., Wang Y.C., Liu C., et al. PD-L1-mediated gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis. Nat. Cell Biol. 2020;22:1264–1275. doi: 10.1038/s41556-020-0575-z. - DOI - PMC - PubMed
-
- Lin C.C., Chin Y.T., Shih Y.J., Chen Y.R., Chung Y.Y., Lin C.Y., Hsiung C.N., Whang-Peng J., Lee S.Y., Lin H.Y., et al. Resveratrol antagonizes thyroid hormone-induced expression of checkpoint and proliferative genes in oral cancer cells. J. Dent. Sci. 2019;14:255–262. doi: 10.1016/j.jds.2019.01.013. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

