Extracellular vesicles as a novel photosensitive drug delivery system for enhanced photodynamic therapy
- PMID: 36237218
- PMCID: PMC9550933
- DOI: 10.3389/fbioe.2022.1032318
Extracellular vesicles as a novel photosensitive drug delivery system for enhanced photodynamic therapy
Abstract
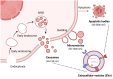
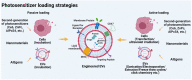
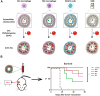
Photodynamic therapy (PDT) is a promising non-invasive therapeutic approach that utilizes photosensitizers (PSs) to generate highly reactive oxygen species (ROS), including singlet oxygen, for removal of targeted cells. PDT has been proven efficacious for the treatment of several diseases, including cancer, cardiovascular disease, inflammatory bowel disease, and diabetic ocular disease. However, the therapeutic efficacy of PDT is limited and often accompanied by side effects, largely due to non-specific delivery of PSs beyond the desired lesion site. Over the past decade, despite various nanoparticular drug delivery systems developed have markedly improved the treatment efficacy while reducing the off-target effects of PSs, concerns over the safety and toxicity of synthetic nanomaterials following intravenous administration are raised. Extracellular vesicles (EVs), a type of nanoparticle released from cells, are emerging as a natural drug delivery system for PSs in light of EV's potentially low immunogenicity and biocompatibility compared with other nanoparticles. This review aims to provide an overview of the research progress in PS delivery systems and propose EVs as an alternative PS delivery system for PDT. Moreover, the challenges and future perspectives of EVs for PS delivery are discussed.
Keywords: drug delivery system; extracellular vesicle; nanoparticle; photodynamic therapy; photosensitizer.
Copyright © 2022 Tong, Zhang, Huang, Yi and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Nanomaterials-based photosensitizers and delivery systems for photodynamic cancer therapy.Biomater Adv. 2022 Apr;135:212725. doi: 10.1016/j.bioadv.2022.212725. Epub 2022 Apr 22. Biomater Adv. 2022. PMID: 35929205 Review.
-
AIE material for photodynamic therapy.Prog Mol Biol Transl Sci. 2021;185:45-73. doi: 10.1016/bs.pmbts.2021.06.010. Epub 2021 Jul 16. Prog Mol Biol Transl Sci. 2021. PMID: 34782107
-
Multifunctional nanoparticles as photosensitizer delivery carriers for enhanced photodynamic cancer therapy.Mater Sci Eng C Mater Biol Appl. 2020 Oct;115:111099. doi: 10.1016/j.msec.2020.111099. Epub 2020 May 16. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 32600703 Review.
-
Progress in Clinical Trials of Photodynamic Therapy for Solid Tumors and the Role of Nanomedicine.Cancers (Basel). 2020 Sep 29;12(10):2793. doi: 10.3390/cancers12102793. Cancers (Basel). 2020. PMID: 33003374 Free PMC article. Review.
-
Nano-photosensitizers for enhanced photodynamic therapy.Photodiagnosis Photodyn Ther. 2021 Dec;36:102597. doi: 10.1016/j.pdpdt.2021.102597. Epub 2021 Oct 24. Photodiagnosis Photodyn Ther. 2021. PMID: 34699982 Review.
Cited by
-
Bacterial Outer Membrane Vesicle (OMV)-Encapsulated TiO2 Nanoparticles: A Dual-Action Strategy for Enhanced Radiotherapy and Immunomodulation in Oral Cancer Treatment.Nanomaterials (Basel). 2024 Dec 20;14(24):2045. doi: 10.3390/nano14242045. Nanomaterials (Basel). 2024. PMID: 39728581 Free PMC article.
-
Extracellular Vesicles as Drug Delivery System for Cancer Therapy.Pharmaceutics. 2024 Aug 1;16(8):1029. doi: 10.3390/pharmaceutics16081029. Pharmaceutics. 2024. PMID: 39204374 Free PMC article. Review.
-
Photodynamic Therapy for Inflammatory and Cancerous Diseases of the Intestines: Molecular Mechanisms and Prospects for Application.Int J Biol Sci. 2023 Sep 4;19(15):4793-4810. doi: 10.7150/ijbs.87492. eCollection 2023. Int J Biol Sci. 2023. PMID: 37781521 Free PMC article. Review.
-
Bacterial outer membrane vesicles as drug delivery carrier for photodynamic anticancer therapy.Front Chem. 2023 Oct 17;11:1284292. doi: 10.3389/fchem.2023.1284292. eCollection 2023. Front Chem. 2023. PMID: 37915541 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

