miR-124-3p sabotages lncRNA MALAT1 stability to repress chondrocyte pyroptosis and relieve cartilage injury in osteoarthritis
- PMID: 36243708
- PMCID: PMC9571420
- DOI: 10.1186/s13018-022-03334-8
miR-124-3p sabotages lncRNA MALAT1 stability to repress chondrocyte pyroptosis and relieve cartilage injury in osteoarthritis
Abstract
Background: Osteoarthritis (OA) is a prevalent inflammatory joint disorder. microRNAs (miRNAs) are increasingly involved in OA.
Aim: Our study is proposed to clarify the role of miR-124-3p in chondrocyte pyroptosis and cartilage injury in OA.
Methods: OA mouse model was established via the treatment of destabilization of the medial meniscus (DMM), and the in vitro cell model was also established as mouse chondrocytes were induced by lipopolysaccharide (LPS). Mouse cartilage injury was assessed using safranin-O-fast green staining, hematoxylin-eosin staining, and OARSI grading method. Expressions of miR-124-3p, MALAT1, KLF5, and CXCL11 were determined. Cartilage injury (MMP-13, osteocalcin), inflammation (IL-6, IL-2, TNF-, IL-1β, and IL-18)- and pyroptosis-related factors (Cleaved Caspase-1 and GSDMD-N) levels were detected. Mechanically, MALAT1 subcellular localization was confirmed. The binding relationships of miR-124-3p and MALAT1 and MALAT1 and KLF5 were verified. MALAT1 half-life period was detected. Then, miR-124-3p was overexpressed using agomiR-124-3p to perform the rescue experiments with oe-MALAT1 or oe-CXCL11.
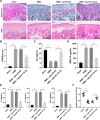
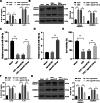
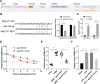
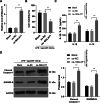
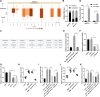
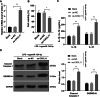
Results: miR-124-3p was downregulated in DMM mice and LPS-induced chondrocytes where cartilage injury, and increased levels of inflammation- and pyroptosis-related factors were found. miR-124-3p overexpression relieved cartilage injury and repressed chondrocyte pyroptosis. miR-124-3p bounds to MALAT1 to downregulate its stability and expression, and MALAT1 bounds to KLF5 to enhance CXCL11 transcription. Overexpression of MALAT1 or CXCL11 annulled the repressive function of miR-124-3p in chondrocyte pyroptosis.
Conclusion: miR-124-3p reduced MALAT1 stability and inhibited the binding of MALAT1 and KLF5 to downregulate CXCL11, thereby suppressing chondrocyte pyroptosis and cartilage injury in OA.
Keywords: Chondrocytes; LncRNA MALAT1; LncRNA stability; Osteoarthritis; Pyroptosis; microRNA-124-3p.
© 2022. The Author(s).
Conflict of interest statement
The authors report there are no competing interests to declare.
Figures
References
-
- Glyn-Jones S, Palmer AJ, Agricola R, Price AJ, Vincent TL, Weinans H, et al. Osteoarthritis Lancet. 2015;386(9991):376–387. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

