A novel microRNA signature for the detection of melanoma by liquid biopsy
- PMID: 36243798
- PMCID: PMC9571479
- DOI: 10.1186/s12967-022-03668-1
A novel microRNA signature for the detection of melanoma by liquid biopsy
Abstract
Background: Melanoma is the deadliest form of skin cancer and metastatic disease is associated with a significant survival rate drop. There is an urgent need for consistent tumor biomarkers to scale precision medicine and reduce cancer mortality. Here, we aimed to identify a melanoma-specific circulating microRNA signature and assess its value as a diagnostic tool.
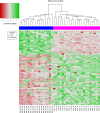
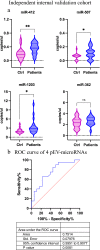
Methods: The study consisted of a discovery phase and two validation phases. Circulating plasma extracellular vesicles (pEV) associated microRNA profiles were obtained from a discovery cohort of metastatic melanoma patients and normal subjects as controls. A pEV-microRNA signature was obtained using a LASSO penalized logistic regression model. The pEV-microRNA signature was subsequently validated both in a publicly available dataset and in an independent internal cohort.
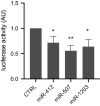
Results: We identified and validated in three independent cohorts a panel of melanoma-specific circulating microRNAs that showed high accuracy in differentiating melanoma patients from healthy subjects with an area under the curve (AUC) of 1.00, 0.94 and 0.75 respectively. Investigation of the function of the pEV-microRNA signature evidenced their possible immune suppressive role in melanoma patients.
Conclusions: We demonstrate that a blood test based on circulating microRNAs can non-invasively detect melanoma, offering a novel diagnostic tool for improving standard care. Moreover, we revealed an immune suppressive role for melanoma pEV-microRNAs.
Keywords: Biomarkers signature; Diagnosis; Extracellular vesicles; Liquid biopsy; Melanoma; microRNAs.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
The authors state no conflict of interest.
Figures


References
-
- Ascierto PA, Dréno B, Larkin J, Ribas A, Liszkay G, Maio M, et al. 5-Year Outcomes with cobimetinib plus vemurafenib in BRAF V600 mutation-positive advanced melanoma: extended follow-up of the coBRIM study. Clin Cancer Res. 2021;27(19):5225–35. doi: 10.1158/1078-0432.CCR-21-0809. - DOI - PMC - PubMed
-
- Ascierto PA, Dummer R, Gogas HJ, Flaherty KT, Arance A, Mandala M, et al. Update on tolerability and overall survival in COLUMBUS: landmark analysis of a randomised phase 3 trial of encorafenib plus binimetinib vs vemurafenib or encorafenib in patients with BRAF V600–mutant melanoma. Eur J Cancer. 2020;126:33–44. doi: 10.1016/j.ejca.2019.11.016. - DOI - PubMed
-
- Robert C, Ribas A, Schachter J, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019;20(9):1239–51. doi: 10.1016/S1470-2045(19)30388-2. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

