The Role of Mitochondrial and Redox Alterations in the Skeletal Myopathy Associated with Chronic Kidney Disease
- PMID: 36245209
- PMCID: PMC9986033
- DOI: 10.1089/ars.2022.0143
The Role of Mitochondrial and Redox Alterations in the Skeletal Myopathy Associated with Chronic Kidney Disease
Abstract
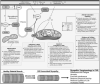
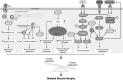
Significance: An estimated 700 million people globally suffer from chronic kidney disease (CKD). In addition to increasing cardiovascular disease risk, CKD is a catabolic disease that results in a loss of muscle mass and function, which are strongly associated with mortality and a reduced quality of life. Despite the importance of muscle health and function, there are no treatments available to prevent or attenuate the myopathy associated with CKD. Recent Advances: Recent studies have begun to unravel the changes in mitochondrial and redox homeostasis within skeletal muscle during CKD. Impairments in mitochondrial metabolism, characterized by reduced oxidative phosphorylation, are found in both rodents and patients with CKD. Associated with aberrant mitochondrial function, clinical and preclinical findings have documented signs of oxidative stress, although the molecular source and species are ill-defined. Critical Issues: First, we review the pathobiology of CKD and its associated myopathy, and we review muscle cell bioenergetics and redox biology. Second, we discuss evidence from clinical and preclinical studies that have implicated the involvement of mitochondrial and redox alterations in CKD-associated myopathy and review the underlying mechanisms reported. Third, we discuss gaps in knowledge related to mitochondrial and redox alterations on muscle health and function in CKD. Future Directions: Despite what has been learned, effective treatments to improve muscle health in CKD remain elusive. Further studies are needed to uncover the complex mitochondrial and redox alterations, including post-transcriptional protein alterations, in patients with CKD and how these changes interact with known or unknown catabolic pathways contributing to poor muscle health and function. Antioxid. Redox Signal. 38, 318-337.
Keywords: cachexia; metabolism; renal; skeletal muscle; uremia.
Conflict of interest statement
The authors have no conflicts, financial or otherwise, to report.
Figures




Similar articles
-
Impaired muscle mitochondrial energetics is associated with uremic metabolite accumulation in chronic kidney disease.JCI Insight. 2020 Dec 8;6(1):e139826. doi: 10.1172/jci.insight.139826. JCI Insight. 2020. PMID: 33290279 Free PMC article.
-
Uremic Myopathy and Mitochondrial Dysfunction in Kidney Disease.Int J Mol Sci. 2022 Nov 4;23(21):13515. doi: 10.3390/ijms232113515. Int J Mol Sci. 2022. PMID: 36362298 Free PMC article.
-
Chronic kidney disease and acquired mitochondrial myopathy.Curr Opin Nephrol Hypertens. 2018 Mar;27(2):113-120. doi: 10.1097/MNH.0000000000000393. Curr Opin Nephrol Hypertens. 2018. PMID: 29266014 Review.
-
Activation of the Aryl Hydrocarbon Receptor in Muscle Exacerbates Ischemic Pathology in Chronic Kidney Disease.Circ Res. 2023 Jul 7;133(2):158-176. doi: 10.1161/CIRCRESAHA.123.322875. Epub 2023 Jun 16. Circ Res. 2023. PMID: 37325935 Free PMC article.
-
New insights into muscle function in chronic kidney disease and metabolic acidosis.Curr Opin Nephrol Hypertens. 2021 May 1;30(3):369-376. doi: 10.1097/MNH.0000000000000700. Curr Opin Nephrol Hypertens. 2021. PMID: 33767065 Review.
Cited by
-
Deletion of the aryl hydrocarbon receptor in endothelial cells improves ischemic angiogenesis in chronic kidney disease.Am J Physiol Heart Circ Physiol. 2024 Jan 1;326(1):H44-H60. doi: 10.1152/ajpheart.00530.2023. Epub 2023 Nov 3. Am J Physiol Heart Circ Physiol. 2024. PMID: 37921663 Free PMC article.
-
Exercise for patients with chronic kidney disease: from cells to systems to function.Am J Physiol Renal Physiol. 2024 Mar 1;326(3):F420-F437. doi: 10.1152/ajprenal.00302.2023. Epub 2024 Jan 11. Am J Physiol Renal Physiol. 2024. PMID: 38205546 Free PMC article. Review.
-
A tryptophan-derived uremic metabolite/Ahr/Pdk4 axis governs skeletal muscle mitochondrial energetics in chronic kidney disease.JCI Insight. 2024 Apr 23;9(10):e178372. doi: 10.1172/jci.insight.178372. JCI Insight. 2024. PMID: 38652558 Free PMC article.
-
Exploring Aluminum Tolerance Mechanisms in Plants with Reference to Rice and Arabidopsis: A Comprehensive Review of Genetic, Metabolic, and Physiological Adaptations in Acidic Soils.Plants (Basel). 2024 Jun 25;13(13):1760. doi: 10.3390/plants13131760. Plants (Basel). 2024. PMID: 38999600 Free PMC article. Review.
-
Temporal serum metabolomic and lipidomic analyses distinguish patients with access-related hand disability following arteriovenous fistula creation.Sci Rep. 2023 Oct 5;13(1):16811. doi: 10.1038/s41598-023-43664-z. Sci Rep. 2023. PMID: 37798334 Free PMC article.
References
-
- Andre M, Huang E, Everly M, et al. The UNOS renal transplant registry: Review of the last decade. Clin Transpl 2014:1–12. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

