Serum glial fibrillary acidic protein and neurofilament light chain in patients with early treated phenylketonuria
- PMID: 36247773
- PMCID: PMC9559705
- DOI: 10.3389/fneur.2022.1011470
Serum glial fibrillary acidic protein and neurofilament light chain in patients with early treated phenylketonuria
Abstract
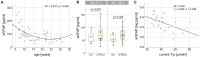
To pave the way for healthy aging in early treated phenylketonuria (ETPKU) patients, a better understanding of the neurological course in this population is needed, requiring easy accessible biomarkers to monitor neurological disease progression in large cohorts. The objective of this pilot study was to investigate the potential of glial fibrillary acidic protein (GFAP) and neurofilament light chain (NfL) as blood biomarkers to indicate changes of the central nervous system in ETPKU. In this single-center cross-sectional study, GFAP and NfL concentrations in serum were quantified using the Simoa® multiplex technology in 56 ETPKU patients aged 6-36 years and 16 age matched healthy controls. Correlation analysis and hierarchical linear regression analysis were performed to investigate an association with disease-related biochemical parameters and retinal layers assessed by optical coherence tomography. ETPKU patients did not show significantly higher GFAP concentrations (mean 73 pg/ml) compared to healthy controls (mean 60 pg/ml, p = 0.140). However, individual pediatric and adult ETPKU patients had GFAP concentrations above the healthy control range. In addition, there was a significant association of GFAP concentrations with current plasma tyrosine concentrations (r = -0.482, p = 0.036), a biochemical marker in phenylketonuria, and the retinal inner nuclear layer volume (r = 0.451, p = 0.04). There was no evidence of NfL alterations in our ETPKU cohort. These pilot results encourage multicenter longitudinal studies to further investigate serum GFAP as a complementary tool to better understand and monitor neurological disease progression in ETPKU. Follow-up investigations on aging ETPKU patients are required to elucidate the potential of serum NfL as biomarker.
Keywords: Simoa assay; biomarkers brain alterations; glial alterations; glial fibrillary acidic protein (GFAP); neuroaxonal damage; neurofilament light chain (NFL); optical coherence tomography; phenylketonuria (PKU).
Copyright © 2022 Lotz-Havla, Katzdobler, Nuscher, Weiß, Levin, Havla and Maier.
Conflict of interest statement
Author AL-H has received travel reimbursement from BioMarin, Nutritia Metabolics, and Sobi and author fees from Thieme medical publishers. Authors JL and SK were funded by the Deutsche Forschungsgemeinschaft (DFG) under Germany's Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy – ID 390857198). Author JL reports speaker fees from Bayer Vital, Biogen and Roche, consulting fees from Axon Neuroscience and Biogen, author fees from Thieme medical publishers and W. Kohlhammer GmbH medical publishers. In addition, he reports compensation for serving as chief medical officer for MODAG GmbH, is beneficiary of the phantom share program of MODAG GmbH and is inventor in a patent Pharmaceutical Composition and Methods of Use (EP 22 159 408.8) filed by MODAG GmbH, all activities outside the submitted work. Author JH is (partially) funded by the German Federal Ministry of Education and Research [Grant Numbers 01ZZ1603[A-D] and 01ZZ1804[A-H] (DIFUTURE)]. Author JH reports a grant for OCT research from the Friedrich-Baur-Stiftung, personal fees and non-financial support from Merck, Alexion, Novartis, Roche, Santhera, Biogen, Heidelberg Engineering, Sanofi Genzyme and non-financial support of the Guthy-Jackson Charitable Foundation, all outside the submitted work. Author EM has received a research grant from Nutritia Metabolic for this work and travel reimbursement from Sobi and Dr. Schär and was paid for advisory services from Sobi, APR and Sanofi-Aventis. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Serum and cerebrospinal fluid neurofilament light chain and glial fibrillary acid protein levels in early and advanced stages of cerebral amyloid Angiopathy.Alzheimers Res Ther. 2024 Apr 23;16(1):86. doi: 10.1186/s13195-024-01457-0. Alzheimers Res Ther. 2024. PMID: 38654326 Free PMC article.
-
Association of serum neurofilament light chain and glial fibrillary acidic protein levels with cognitive decline in Parkinson's disease.Brain Res. 2023 Apr 15;1805:148271. doi: 10.1016/j.brainres.2023.148271. Epub 2023 Feb 7. Brain Res. 2023. PMID: 36754139
-
Serum glial fibrillary acidic protein and neurofilament light chain in treatment-naïve patients with unipolar depression.J Affect Disord. 2023 Oct 1;338:341-348. doi: 10.1016/j.jad.2023.06.028. Epub 2023 Jun 17. J Affect Disord. 2023. PMID: 37336248
-
Reduced macular thickness and macular vessel density in early-treated adult patients with PKU.Mol Genet Metab Rep. 2021 May 5;27:100767. doi: 10.1016/j.ymgmr.2021.100767. eCollection 2021 Jun. Mol Genet Metab Rep. 2021. PMID: 34026550 Free PMC article.
-
Biomarkers in autoimmune diseases of the central nervous system.Front Immunol. 2023 Apr 5;14:1111719. doi: 10.3389/fimmu.2023.1111719. eCollection 2023. Front Immunol. 2023. PMID: 37090723 Free PMC article. Review.
References
-
- Muntau AC, Burlina A, Eyskens F, Freisinger P, De Laet C, Leuzzi V, et al. . Efficacy, safety and population pharmacokinetics of sapropterin in pku patients < 4 years: results from the spark open-label, multicentre, randomized phase iiib trial. Orphanet J Rare Dis. (2017) 12:47. 10.1186/s13023-017-0600-x - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

