Structural changes in the retina after implantation of subretinal three-dimensional implants in mini pigs
- PMID: 36248640
- PMCID: PMC9561346
- DOI: 10.3389/fnins.2022.1010445
Structural changes in the retina after implantation of subretinal three-dimensional implants in mini pigs
Abstract
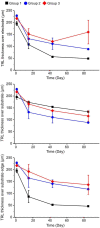
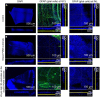
The retinal structural changes after subretinal implantation of three-dimensional (3D) microelectrodes were investigated in a mini pig. Three types of electrode were implanted into the subretinal spaces of nine mini pigs: 75-μm-high 3D electrodes on a 200-μm-thick right-angled polydimethylsiloxane (PDMS) substrate (group 1); a 140-μm-thick sloped PDMS substrate without electrodes (group 2); and a 140-μm-thick sloped PDMS substrate with 20-μm-high 3D electrodes (group 3). One mini pig was used as a control. Spectral domain-optical coherence tomography (SD-OCT) images were obtained at baseline and 2, 6, and 12 weeks post-surgery. Retinal specimens were immunostained using a tissue-clearing method 3 months post-implantation. The 75-μm-high 3D electrodes progressively penetrated the inner nuclear layer (INL) and touched the inner plexiform layer (IPL) 2 weeks post-surgery. At 6 weeks post-operatively, the electrodes were in contact with the nerve-fiber layer, accompanied by a severe fibrous reaction. In the other groups, the implants remained in place without subretinal migration. Immunostaining showed that retinal ganglion and bipolar cells were preserved without fibrosis over the retinal implants in groups 2 and 3 during the 12-week implantation period. In summary, SD-OCT and immunohistology results showed differences in the extent of reactions, such as fibrosis over the implants and penetration of the electrodes into the inner retinal layer depending on different types of electrodes. A sloped substrate performed better than a right-angled substrate in terms of retinal preservation over the implanted electrodes. The 20-μm-high electrodes showed better structural compatibility than the 75-μm-high 3D electrodes. There was no significant difference between the results of sloped implants without electrodes and 20-μm-high 3D electrodes, indicating that the latter had no adverse effects on retinal tissue.
Keywords: implant design; retinal prosthesis; structural retinal change; subretinal implant; three-dimensional microelectrodes.
Copyright © 2022 Vu, Seo, Choi, Kim, Kang, Lee, Park, Kim, Kim and Kim.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
LinkOut - more resources
Full Text Sources

