Implications of Porphyromonas gingivalis peptidyl arginine deiminase and gingipain R in human health and diseases
- PMID: 36250046
- PMCID: PMC9559808
- DOI: 10.3389/fcimb.2022.987683
Implications of Porphyromonas gingivalis peptidyl arginine deiminase and gingipain R in human health and diseases
Abstract
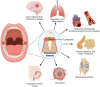
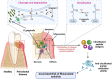
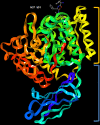
Porphyromonas gingivalis is a major pathogenic bacterium involved in the pathogenesis of periodontitis. Citrullination has been reported as the underlying mechanism of the pathogenesis, which relies on the interplay between two virulence factors of the bacterium, namely gingipain R and the bacterial peptidyl arginine deiminase. Gingipain R cleaves host proteins to expose the C-terminal arginines for peptidyl arginine deiminase to citrullinate and generate citrullinated proteins. Apart from carrying out citrullination in the periodontium, the bacterium is found capable of citrullinating proteins present in the host synovial tissues, atherosclerotic plaques and neurons. Studies have suggested that both virulence factors are the key factors that trigger distal effects mediated by citrullination, leading to the development of some non-communicable diseases, such as rheumatoid arthritis, atherosclerosis, and Alzheimer's disease. Thus, inhibition of these virulence factors not only can mitigate periodontitis, but also can provide new therapeutic solutions for systematic diseases involving bacterial citrullination. Herein, we described both these proteins in terms of their unique structural conformations and biological relevance to different human diseases. Moreover, investigations of inhibitory actions on the enzymes are also enumerated. New approaches for identifying inhibitors for peptidyl arginine deiminase through drug repurposing and virtual screening are also discussed.
Keywords: Porphyromonas gingivalis; citrullination; gingipain R; inhibitors; peptidyl arginine deiminase; systemic disease.
Copyright © 2022 Chow, Yam, Gunasekaran, Lai, Wo, Agarwal, Ong, Cheong and Tan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Peptidylarginine deiminase from Porphyromonas gingivalis citrullinates human fibrinogen and α-enolase: implications for autoimmunity in rheumatoid arthritis.Arthritis Rheum. 2010 Sep;62(9):2662-72. doi: 10.1002/art.27552. Arthritis Rheum. 2010. PMID: 20506214 Free PMC article.
-
A Secreted Bacterial Peptidylarginine Deiminase Can Neutralize Human Innate Immune Defenses.mBio. 2018 Oct 30;9(5):e01704-18. doi: 10.1128/mBio.01704-18. mBio. 2018. PMID: 30377277 Free PMC article.
-
Extracellular Proteome and Citrullinome of the Oral Pathogen Porphyromonas gingivalis.J Proteome Res. 2016 Dec 2;15(12):4532-4543. doi: 10.1021/acs.jproteome.6b00634. Epub 2016 Oct 19. J Proteome Res. 2016. PMID: 27712078
-
Periodontal sources of citrullinated antigens and TLR agonists related to RA.Autoimmunity. 2018 Sep;51(6):304-309. doi: 10.1080/08916934.2018.1527907. Epub 2018 Nov 10. Autoimmunity. 2018. PMID: 30417696 Review.
-
Citrullination as a plausible link to periodontitis, rheumatoid arthritis, atherosclerosis and Alzheimer's disease.J Oral Microbiol. 2018 Jun 22;10(1):1487742. doi: 10.1080/20002297.2018.1487742. eCollection 2018. J Oral Microbiol. 2018. PMID: 29963294 Free PMC article. Review.
Cited by
-
Smoking Impacts Alzheimer's Disease Progression Through Oral Microbiota Modulation.Mol Neurobiol. 2025 Jan;62(1):19-44. doi: 10.1007/s12035-024-04241-1. Epub 2024 May 25. Mol Neurobiol. 2025. PMID: 38795302 Review.
-
Porphyromonas gingivalis Vaccine: Antigens and Mucosal Adjuvants.Vaccines (Basel). 2024 Jun 4;12(6):619. doi: 10.3390/vaccines12060619. Vaccines (Basel). 2024. PMID: 38932348 Free PMC article. Review.
-
In Silico Docking of Medicinal Herbs Against P. gingivalis for Chronic Periodontitis Intervention.Int Dent J. 2025 Apr;75(2):1113-1135. doi: 10.1016/j.identj.2024.06.019. Epub 2024 Aug 10. Int Dent J. 2025. PMID: 39127518 Free PMC article.
-
Unveiling the Molecular Crosstalk Between Periodontal and Cardiovascular Diseases: A Systematic Review.Dent J (Basel). 2025 Feb 25;13(3):98. doi: 10.3390/dj13030098. Dent J (Basel). 2025. PMID: 40136726 Free PMC article. Review.
-
Who is in the driver's seat? Parvimonas micra: An understudied pathobiont at the crossroads of dysbiotic disease and cancer.Environ Microbiol Rep. 2023 Aug;15(4):254-264. doi: 10.1111/1758-2229.13153. Epub 2023 Mar 30. Environ Microbiol Rep. 2023. PMID: 36999244 Free PMC article. Review.
References
-
- Abe N., Kadowaki T., Okamoto K., Nakayama K., Ohishi M., Yamamoto K. (1998). Biochemical and functional properties of lysine-specific cysteine proteinase (Lys-gingipain) as a virulence factor of porphyromonas gingivalis in periodontal disease. J. Biochem. 123 (2), 305–312. doi: 10.1093/oxfordjournals.jbchem.a021937 - DOI - PubMed
-
- Alhogail S., Suaifan G., Bizzarro S., Kaman W. E., Bikker F. J., Weber K., et al. . (2018). On site visual detection of porphyromonas gingivalis related periodontitis by using a magnetic-nanobead based assay for gingipains protease biomarkers. Microchim. Acta 185 (2), 149. doi: 10.1007/s00604-018-2677-x - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

