Regulated Restructuring of Mucins During Secretory Granule Maturation In Vivo
- PMID: 36252017
- PMCID: PMC9618048
- DOI: 10.1073/pnas.2209750119
Regulated Restructuring of Mucins During Secretory Granule Maturation In Vivo
Abstract
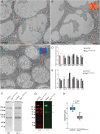
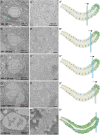
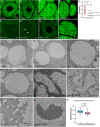
Mucins are large, highly glycosylated transmembrane and secreted proteins that line and protect epithelial surfaces. However, the details of mucin biosynthesis and packaging in vivo are largely unknown. Here, we demonstrate that multiple distinct mucins undergo intragranular restructuring during secretory granule maturation in vivo, forming unique structures that are spatially segregated within the same granule. We further identify temporally-regulated genes that influence mucin restructuring, including those controlling pH (Vha16-1), Ca2+ ions (fwe) and Cl- ions (Clic and ClC-c). Finally, we show that altered mucin glycosylation influences the dimensions of these structures, thereby affecting secretory granule morphology. This study elucidates key steps and factors involved in intragranular, rather than intergranular segregation of mucins through regulated restructuring events during secretory granule maturation. Understanding how multiple distinct mucins are efficiently packaged into and secreted from secretory granules may provide insight into diseases resulting from defects in mucin secretion.
Keywords: O-glycosylation; mucin; salivary gland; secretion; secretory granules.
Conflict of interest statement
The authors declare no competing interest.
Figures


Similar articles
-
A novel cysteine-rich adaptor protein is required for mucin packaging and secretory granule stability in vivo.Proc Natl Acad Sci U S A. 2024 Feb 6;121(6):e2314309121. doi: 10.1073/pnas.2314309121. Epub 2024 Jan 29. Proc Natl Acad Sci U S A. 2024. PMID: 38285943 Free PMC article.
-
Mucin granule intraluminal organization in living mucous/goblet cells. Roles of protein post-translational modifications and secretion.J Biol Chem. 2006 Feb 24;281(8):4844-55. doi: 10.1074/jbc.M510520200. Epub 2005 Dec 23. J Biol Chem. 2006. PMID: 16377632
-
A novel function for Rab1 and Rab11 during secretory granule maturation.J Cell Sci. 2021 Aug 1;134(15):jcs259037. doi: 10.1242/jcs.259037. Epub 2021 Aug 3. J Cell Sci. 2021. PMID: 34342349 Free PMC article.
-
Mucin granule intraluminal organization.Am J Respir Cell Mol Biol. 2007 Feb;36(2):183-90. doi: 10.1165/rcmb.2006-0291TR. Epub 2006 Sep 7. Am J Respir Cell Mol Biol. 2007. PMID: 16960124 Free PMC article. Review.
-
In vivo models of mucin biosynthesis and function.Adv Drug Deliv Rev. 2022 May;184:114182. doi: 10.1016/j.addr.2022.114182. Epub 2022 Mar 9. Adv Drug Deliv Rev. 2022. PMID: 35278522 Free PMC article. Review.
Cited by
-
O-Glycosylation of a male seminal fluid protein influences sperm binding and female postmating behavior.PNAS Nexus. 2024 Aug 6;3(8):pgae322. doi: 10.1093/pnasnexus/pgae322. eCollection 2024 Aug. PNAS Nexus. 2024. PMID: 39189023 Free PMC article.
-
A novel cysteine-rich adaptor protein is required for mucin packaging and secretory granule stability in vivo.Proc Natl Acad Sci U S A. 2024 Feb 6;121(6):e2314309121. doi: 10.1073/pnas.2314309121. Epub 2024 Jan 29. Proc Natl Acad Sci U S A. 2024. PMID: 38285943 Free PMC article.
-
The multifaceted roles of mucins family in lung cancer: from prognostic biomarkers to promising targets.Front Immunol. 2025 Jun 27;16:1608140. doi: 10.3389/fimmu.2025.1608140. eCollection 2025. Front Immunol. 2025. PMID: 40655139 Free PMC article. Review.
-
The Ykt6-Snap29-Syx13 SNARE complex promotes crinophagy via secretory granule fusion with Lamp1 carrier vesicles.Sci Rep. 2024 Feb 8;14(1):3200. doi: 10.1038/s41598-024-53607-x. Sci Rep. 2024. PMID: 38331993 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

