Gastric-type adenocarcinoma of the cervix: Clinical outcomes and genomic drivers
- PMID: 36253302
- PMCID: PMC10155605
- DOI: 10.1016/j.ygyno.2022.10.003
Gastric-type adenocarcinoma of the cervix: Clinical outcomes and genomic drivers
Abstract
Objectives: Gastric-type endocervical adenocarcinoma (GEA) is a rare form of cervical cancer not associated with human papilloma virus (HPV) infection. We summarize our experience with GEA at a large cancer center.
Methods: Clinical and demographic information on all patients diagnosed with GEA between June 1, 2002 and July 1, 2019 was obtained retrospectively from clinical charts. Kaplan-Meier survival analysis was performed to describe progression-free survival (PFS) and overall survival (OS). Tumors from a subset of patients underwent next generation sequencing (NGS) analysis.
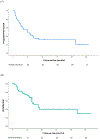
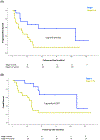
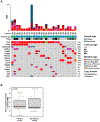
Results: A total of 70 women with GEA were identified, including 43 who received initial treatment at our institution: of these 4 (9%) underwent surgery alone, 15 (35%) underwent surgery followed by adjuvant therapy, 10 (23%) were treated with definitive concurrent chemoradiation (CCRT), 7 (16%) with chemotherapy alone, and 3 (7%) with neoadjuvant CCRT and hysterectomy with or without chemotherapy. One-third (n = 14) of patients experienced disease progression, of whom 86% (n = 12) had prior CCRT. The median PFS and OS for patients with stage I GEA were 107 months (95% CI 14.8-199.2 months) and 111 months (95% CI 17-205.1 months) respectively, compared to 17 months (95% CI 5.6-28.4 months) and 33 months (95% CI 28.2-37.8 months) for patients with stages II-IV, respectively. On NGS, 4 patients (14%) had ERBB2 alterations, including 2 patients who received trastuzumab.
Conclusions: GEA is an aggressive form of cervical cancer with poor PFS and OS when diagnosed at stage II or later. Further investigation is needed to identify the optimal management approach for this rare subtype.
Keywords: Adenoma malignum; Cervical cancer; Endocervical cancer; Gastric-type endocervical adenocarcinoma; Genetic profile.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest Outside the submitted work, B. Weigelt reports ad hoc membership in the scientific advisory board of Repare Therapeutics, outside the scope of the study. C. Friedman reports participation in the scientific advisory boards for Merck (LYNK-002) and Genentech (MyPathway) without compensation, consulting for Seagen and Bristol Myers Squibb, and institutional research funds from Genentech/Roche, Bristol Myers Squibb, Merck, AstraZeneca, and Daiichi. E. Jewell reports personal fee from Covidien/Medtronic. K. Cadoo reports grant funding from the Irish Cancer Society, MSD, and Immunogen; consulting fees from Nextcure, MJH Life Sciences, and GSK; payments/honoraria from GSK, AstraZeneca, and MSD; financial support to attend meetings from Roche, Pfizer, and MSD; advisory board participation at MSD, AstraZeneca, GSK, and Eisai; a voluntary advisory role at the National Cancer Control Programme Ireland; and a voluntary board member at ARC Cancer Support Centers. N. Abu-Rustum reports grant funding from GRAIL paid to the institution. S. issa Bhaloo reports stock or stock options in BioNTech, Moderna, Inc., Inovio, Relief Therapeutics Holding SA, Cansino Biologics, Inc., and Pfizer, Inc. M. Leitao reports personal fees from Medtronic, Intuitive Surgical, Inc., and JnJ/Ethicon. C. Aghajanian has received research grants from Abbvie, Clovis, Genentech, and Astra Zeneca and served on advisory boards for Abbvie, AstraZeneca/Merck, Eisai/Merck, Mersana Therapeutics, Repare Therapeutics, and Roche/Genentech. R. O'Cearbhaill reports honoraria from GSK, Bayer, Regeneron, SeaGen, Fresenius Kabi, Immunogen, MJH Life Sciences and Curio. C. Kyi reports grant funding from Conquer Cancer Foundation; grant funding paid to the institution from Merus, Gritstone, and BMS; and consulting fees from Scenic Immunology B.V. and OncLive. D. Zamarin is an inventor on a patent related to the use of oncolytic Newcastle Disease Virus for cancer therapy, and reports grant funding paid to the institution from AstraZeneca, Roche, Plexxikon, and Synthekine; consulting fees from Memgen, Celldex, Agenus, Astellas, AstraZeneca, Crown Biosciences, Roche, GSK, Hookpia, ImmunOS, Kalvir, Synlogic Therapeutics, Synthekine, Takeda, Targovax, Tessa Therapeutics, and Xencor; stock options in Accurius Therapeutics, Immunos Therapeutics and Calidi Biotherapeutics; and a Merck licensed patent (with personal payments and payments to the institution). All other authors have no potential conflicts of interest to disclose.
Figures

References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. - PubMed
-
- Smith HO, Tiffany MF, Qualls CR, Key CR. The rising incidence of adenocarcinoma relative to squamous cell carcinoma of the uterine cervix in the United States--a 24-year population-based study. Gynecol Oncol. 2000;78:97–105. - PubMed
-
- Hodgson A, Park KJ. Cervical Adenocarcinomas: A Heterogeneous Group of Tumors With Variable Etiologies and Clinical Outcomes. Arch Pathol Lab Med. 2019;143:34–46. - PubMed
-
- Park KJ. Cervical adenocarcinoma: integration of HPV status, pattern of invasion, morphology and molecular markers into classification. Histopathology. 2020;76:112–27. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

