Reduced penetrance of MODY-associated HNF1A/HNF4A variants but not GCK variants in clinically unselected cohorts
- PMID: 36257325
- PMCID: PMC9674944
- DOI: 10.1016/j.ajhg.2022.09.014
Reduced penetrance of MODY-associated HNF1A/HNF4A variants but not GCK variants in clinically unselected cohorts
Abstract
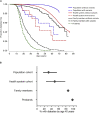
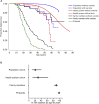
The true prevalence and penetrance of monogenic disease variants are often not known because of clinical-referral ascertainment bias. We comprehensively assess the penetrance and prevalence of pathogenic variants in HNF1A, HNF4A, and GCK that account for >80% of monogenic diabetes. We analyzed clinical and genetic data from 1,742 clinically referred probands, 2,194 family members, clinically unselected individuals from a US health system-based cohort (n = 132,194), and a UK population-based cohort (n = 198,748). We show that one in 1,500 individuals harbor a pathogenic variant in one of these genes. The penetrance of diabetes for HNF1A and HNF4A pathogenic variants was substantially lower in the clinically unselected individuals compared to clinically referred probands and was dependent on the setting (32% in the population, 49% in the health system cohort, 86% in a family member, and 98% in probands for HNF1A). The relative risk of diabetes was similar across the clinically unselected cohorts highlighting the role of environment/other genetic factors. Surprisingly, the penetrance of pathogenic GCK variants was similar across all cohorts (89%-97%). We highlight that pathogenic variants in HNF1A, HNF4A, and GCK are not ultra-rare in the population. For HNF1A and HNF4A, we need to tailor genetic interpretation and counseling based on the setting in which a pathogenic monogenic variant was identified. GCK is an exception with near-complete penetrance in all settings. This along with the clinical implication of diagnosis makes it an excellent candidate for the American College of Medical Genetics secondary gene list.
Keywords: ACMG; GCK; HNF1A; HNF4A; MODY; monogenic diabetes; monogenic disease; penetrance; population screening; prevalence; variant curation.
Copyright © 2022 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures
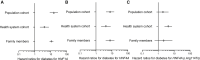

References
-
- McDonald T.J., Ellard S. Maturity onset diabetes of the young: identification and diagnosis. Ann. Clin. Biochem. 2013;50:403–415. - PubMed
-
- Pearson E.R., Starkey B.J., Powell R.J., Gribble F.M., Clark P.M., Hattersley A.T. Genetic cause of hyperglycaemia and response to treatment in diabetes. Lancet. 2003;362:1275–1281. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

