Neurological autoimmune diseases following vaccinations against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): A follow-up study
- PMID: 36259114
- PMCID: PMC9874608
- DOI: 10.1111/ene.15602
Neurological autoimmune diseases following vaccinations against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): A follow-up study
Abstract
Background and purpose: Population-based studies suggest severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines may trigger neurological autoimmunity including immune-mediated thrombotic thrombocytopenia. Long-term characterization of cases is warranted to facilitate patient care and inform vaccine-hesitant individuals.
Methods: In this single-center prospective case study with a median follow-up of 387 days long-term clinical, laboratory and imaging characteristics of patients with neurological autoimmunity diagnosed in temporal association (≤6 weeks) with SARS-CoV-2 vaccinations are reported.
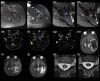
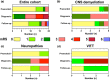
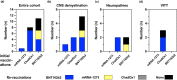
Results: Follow-up data were available for 20 cases (central nervous system demyelinating diseases n = 8, inflammatory peripheral neuropathies n = 4, vaccine-induced immune thrombotic thrombocytopenia n = 3, myositis n = 2, myasthenia n = 1, limbic encephalitis n = 1, giant cell arteritis n = 1). Following therapy, the overall disability level improved (median modified Rankin Scale at diagnosis 3 vs. 1 at follow-up). The condition of two patients worsened despite immunosuppressants possibly related to their autoimmune diagnoses (limbic encephalitis n = 1, giant cell arteritis n = 1). At 12 months' follow-up, 12 patients achieved complete clinical remissions with partial responses in five and stable disease in one case. Correspondingly, autoimmune antibodies were non-detectable or titers had significantly lowered in all, and repeat imaging revealed radiological responses in most cases. Under vigilant monitoring 15 patients from our cohort underwent additional SARS-CoV-2 vaccinations (BNT162b2 n = 12, mRNA-1273 n = 3). Most patients (n = 11) received different vaccines than prior to diagnosis of neurological autoimmunity. Except for one short-lasting relapse, which responded well to steroids, re-vaccinations were well tolerated.
Conclusions: In this study long-term characteristics of neurological autoimmunity encountered after SARS-CoV-2 vaccinations are defined. Outcome was favorable in most cases. Re-vaccinations were well tolerated and should be considered on an individual risk/benefit analysis.
Keywords: COVID-19; Guillain-Barré syndrome; autoimmune; cerebral venous sinus thrombosis; multiple sclerosis; myelitis; myositis.
© 2022 The Authors. European Journal of Neurology published by John Wiley & Sons Ltd on behalf of European Academy of Neurology.
Conflict of interest statement
None declared.
Figures
Comment in
-
Severe acute respiratory syndrome coronavirus 2 vaccines and neurological diseases: Cause or coincidence?Eur J Neurol. 2023 Feb;30(2):303-304. doi: 10.1111/ene.15618. Epub 2022 Nov 9. Eur J Neurol. 2023. PMID: 36318268 Free PMC article. No abstract available.
Similar articles
-
Neurological autoimmune diseases following vaccinations against SARS-CoV-2: a case series.Eur J Neurol. 2022 Feb;29(2):555-563. doi: 10.1111/ene.15147. Epub 2021 Oct 31. Eur J Neurol. 2022. PMID: 34668274 Free PMC article.
-
Type 1 diabetes mellitus following SARS-CoV-2 mRNA vaccination.Endocrine. 2022 Oct;78(1):42-46. doi: 10.1007/s12020-022-03130-8. Epub 2022 Jul 9. Endocrine. 2022. PMID: 35809159 Free PMC article.
-
Seropositive Neuromyelitis Optica in a Case of Undiagnosed Ankylosing Spondylitis: A Neuro-Rheumatological Conundrum.Qatar Med J. 2022 Jul 7;2022(3):29. doi: 10.5339/qmj.2022.29. eCollection 2022. Qatar Med J. 2022. PMID: 35864917 Free PMC article.
-
MOG encephalomyelitis after vaccination against severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2): case report and comprehensive review of the literature.J Neurol. 2022 Oct;269(10):5198-5212. doi: 10.1007/s00415-022-11194-9. Epub 2022 Jun 23. J Neurol. 2022. PMID: 35737110 Free PMC article. Review.
-
Immune-mediated encephalitis following SARS-CoV-2 vaccinations.Clin Neurol Neurosurg. 2024 Mar;238:108188. doi: 10.1016/j.clineuro.2024.108188. Epub 2024 Feb 21. Clin Neurol Neurosurg. 2024. PMID: 38422745 Review.
Cited by
-
Three Cases of Arteritic Anterior Optic Neuropathy Several Months after COVID-19 Vaccination.Case Rep Ophthalmol Med. 2023 Sep 11;2023:8845850. doi: 10.1155/2023/8845850. eCollection 2023. Case Rep Ophthalmol Med. 2023. PMID: 37727533 Free PMC article.
-
Increased incidence of giant cell arteritis and associated stroke during the COVID-19 pandemic in Spain: A nation-wide population study.Autoimmun Rev. 2023 Jun;22(6):103341. doi: 10.1016/j.autrev.2023.103341. Epub 2023 Apr 14. Autoimmun Rev. 2023. PMID: 37062441 Free PMC article. Review.
-
Autoantibodies Targeting G-Protein-Coupled Receptors and RAS-Related Molecules in Post-Acute COVID Vaccination Syndrome: A Retrospective Case Series Study.Biomedicines. 2024 Dec 15;12(12):2852. doi: 10.3390/biomedicines12122852. Biomedicines. 2024. PMID: 39767757 Free PMC article.
-
Severe acute respiratory syndrome coronavirus 2 vaccines and neurological diseases: Cause or coincidence?Eur J Neurol. 2023 Feb;30(2):303-304. doi: 10.1111/ene.15618. Epub 2022 Nov 9. Eur J Neurol. 2023. PMID: 36318268 Free PMC article. No abstract available.
-
The Set up and the Triggers: An Update on the Risk Factors for Giant Cell Arteritis.Curr Neurol Neurosci Rep. 2024 Dec 14;25(1):11. doi: 10.1007/s11910-024-01386-3. Curr Neurol Neurosci Rep. 2024. PMID: 39673667 Review.
References
-
- COVID‐19 Data Repository by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University Available from: https://github.com/CSSEGISandData/COVID‐19.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

