Germline Testing and Somatic Tumor Testing for BRCA1/2 Pathogenic Variants in Ovarian Cancer: What Is the Optimal Sequence of Testing?
- PMID: 36265114
- PMCID: PMC9616645
- DOI: 10.1200/PO.22.00033
Germline Testing and Somatic Tumor Testing for BRCA1/2 Pathogenic Variants in Ovarian Cancer: What Is the Optimal Sequence of Testing?
Abstract
Purpose: In 2020, ASCO recommended that all women with epithelial ovarian cancer have germline testing for BRCA1/2 mutations, and those without a germline pathogenic variant (PV) should have somatic tumor testing to determine eligibility for a poly (ADP-ribose) polymerase inhibitor. Consequently, the majority of patients with ovarian cancer will have both germline testing and somatic testing. An alternate strategy is tumor testing first and then germline testing if there is a PV in the tumor and/or significant family history. The objective was to conduct a cost-effectiveness analysis comparing the two testing strategies.
Methods: The Markov model compared the costs (US dollars) and benefits of two testing strategies for newly diagnosed ovarian cancer: (1) ASCO strategy and (2) tumor testing triage for germline testing. Data were applied from SOLO-1, and costs were from wholesale acquisition prices, Medicare, and published sources. Sensitivity analyses accounted for uncertainty around various parameters. Monte Carlo simulation estimated the number tested and identified with germline and somatic BRCA PV for olaparib maintenance treatment annually in the US population.
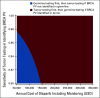
Results: The ASCO strategy was more effective but more costly than tumor testing triage in identifying patients for olaparib, with an incremental cost-effectiveness ratio of $281,296 US dollars per progression-free life year gained. Assuming 10,000 eligible patients with ovarian cancer annually, Monte Carlo simulation yielded comparable numbers of patients with BRCA PV in the germline and tumor with the ASCO and tumor testing triage strategies (2,080 v 2,062, respectively), but substantially higher number of patients tested using the ASCO strategy (8,052 v 3,076).
Conclusion: The ASCO strategy may identify more BRCA PVs but is not cost-effective. Tumor testing in epithelial ovarian cancer as triage for germline testing is the favored strategy in this health care system.
Conflict of interest statement
<b>Janice S. Kwon</b><b>Honoraria:</b> AstraZeneca<b>Consulting or Advisory Role:</b> AstraZeneca<b>Research Funding:</b> AstraZeneca (Inst) <b>Anna V. Tinker</b><b>Honoraria:</b> AstraZeneca, GlaxoSmithKline Canada, Eisai, Viatris, Merck Serono<b>Consulting or Advisory Role:</b> AstraZeneca<b>Research Funding:</b> AstraZeneca/MedImmune (Inst)<b>Other Relationship:</b> AstraZeneca<b>Uncompensated Relationships:</b> GlaxoSmithKline <b>Katie Compton</b><b>Consulting or Advisory Role:</b> AstraZeneca<b>Research Funding:</b> AstraZeneca <b>Sophie Sun</b><b>Consulting or Advisory Role:</b> AstraZeneca, Merck, Novartis <b>Kasmintan A. Schrader</b><b>Consulting or Advisory Role:</b> Pfizer, AstraZeneca, Precision RxDx<b>Research Funding:</b> AstraZeneca (Inst) <b>Aly Karsan</b><b>Honoraria:</b> Jazz Pharmaceuticals<b>Research Funding:</b> AstraZeneca, PfizerNo other potential conflicts of interest were reported.
Figures


Similar articles
-
Cost-Effectiveness of Unselected Multigene Germline and Somatic Genetic Testing for Epithelial Ovarian Cancer.J Natl Compr Canc Netw. 2024 Apr 18;22(2 D):e237331. doi: 10.6004/jnccn.2023.7331. J Natl Compr Canc Netw. 2024. PMID: 38866043
-
The most efficient and effective BRCA1/2 testing strategy in epithelial ovarian cancer: Tumor-First or Germline-First?Gynecol Oncol. 2023 Jul;174:121-128. doi: 10.1016/j.ygyno.2023.04.029. Epub 2023 May 12. Gynecol Oncol. 2023. PMID: 37182432
-
Prevalence of germline pathogenic BRCA1/2 variants in sequential epithelial ovarian cancer cases.J Med Genet. 2019 May;56(5):301-307. doi: 10.1136/jmedgenet-2018-105792. Epub 2019 Jan 25. J Med Genet. 2019. PMID: 30683677
-
An update on current and emerging therapies for epithelial ovarian cancer: Focus on poly(adenosine diphosphate-ribose) polymerase inhibition and antiangiogenesis.J Oncol Pharm Pract. 2017 Sep;23(6):454-469. doi: 10.1177/1078155216657165. Epub 2016 Jun 29. J Oncol Pharm Pract. 2017. PMID: 27357817 Review.
-
Germline and Somatic Tumor Testing in Epithelial Ovarian Cancer: ASCO Guideline.J Clin Oncol. 2020 Apr 10;38(11):1222-1245. doi: 10.1200/JCO.19.02960. Epub 2020 Jan 27. J Clin Oncol. 2020. PMID: 31986064 Free PMC article.
Cited by
-
BRCA1/2 Testing Landscape in Ovarian Cancer: A Nationwide, Real-World Data Study.Cancers (Basel). 2024 Apr 26;16(9):1682. doi: 10.3390/cancers16091682. Cancers (Basel). 2024. PMID: 38730634 Free PMC article.
-
Is Reflex Germline BRCA1/2 Testing Necessary in Women Diagnosed with Non-Mucinous High-Grade Epithelial Ovarian Cancer Aged 80 Years or Older?Cancers (Basel). 2023 Jan 25;15(3):730. doi: 10.3390/cancers15030730. Cancers (Basel). 2023. PMID: 36765687 Free PMC article.
-
Untapped Potential of Poly(ADP-Ribose) Polymerase Inhibitors: Lessons Learned From the Real-World Clinical Homologous Recombination Repair Mutation Testing.World J Oncol. 2024 Aug;15(4):562-578. doi: 10.14740/wjon1820. Epub 2024 Jun 11. World J Oncol. 2024. PMID: 38993246 Free PMC article.
-
Real-World Concordance between Germline and Tumour BRCA1/2 Status in Epithelial Ovarian Cancer.Cancers (Basel). 2023 Dec 29;16(1):177. doi: 10.3390/cancers16010177. Cancers (Basel). 2023. PMID: 38201604 Free PMC article.
References
-
- Banerjee S, Moore KN, Colombo N, et al. : Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-Year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 22:1721-1731, 2021 - PubMed
-
- Moore K, Colombo N, Scambia G, et al. : Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med 379:2495-2505, 2018 - PubMed
-
- Lau TTY, May CM, Sefid Dashti ZJ, et al. : Use of treatment-focused tumor sequencing to screen for germline cancer predisposition. J Mol Diagn 23:1145-1158, 2021 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

