Improved upper limb function in non-ambulant children with SMA type 2 and 3 during nusinersen treatment: a prospective 3-years SMArtCARE registry study
- PMID: 36274155
- PMCID: PMC9589836
- DOI: 10.1186/s13023-022-02547-8
Improved upper limb function in non-ambulant children with SMA type 2 and 3 during nusinersen treatment: a prospective 3-years SMArtCARE registry study
Abstract
Background: The development and approval of disease modifying treatments have dramatically changed disease progression in patients with spinal muscular atrophy (SMA). Nusinersen was approved in Europe in 2017 for the treatment of SMA patients irrespective of age and disease severity. Most data on therapeutic efficacy are available for the infantile-onset SMA. For patients with SMA type 2 and type 3, there is still a lack of sufficient evidence and long-term experience for nusinersen treatment. Here, we report data from the SMArtCARE registry of non-ambulant children with SMA type 2 and typen 3 under nusinersen treatment with a follow-up period of up to 38 months.
Methods: SMArtCARE is a disease-specific registry with data on patients with SMA irrespective of age, treatment regime or disease severity. Data are collected during routine patient visits as real-world outcome data. This analysis included all non-ambulant patients with SMA type 2 or 3 below 18 years of age before initiation of treatment. Primary outcomes were changes in motor function evaluated with the Hammersmith Functional Motor Scale Expanded (HFMSE) and the Revised Upper Limb Module (RULM).
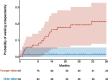
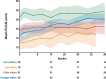
Results: Data from 256 non-ambulant, pediatric patients with SMA were included in the data analysis. Improvements in motor function were more prominent in upper limb: 32.4% of patients experienced clinically meaningful improvements in RULM and 24.6% in HFMSE. 8.6% of patients gained a new motor milestone, whereas no motor milestones were lost. Only 4.3% of patients showed a clinically meaningful worsening in HFMSE and 1.2% in RULM score.
Conclusion: Our results demonstrate clinically meaningful improvements or stabilization of disease progression in non-ambulant, pediatric patients with SMA under nusinersen treatment. Changes were most evident in upper limb function and were observed continuously over the follow-up period. Our data confirm clinical trial data, while providing longer follow-up, an increased number of treated patients, and a wider range of age and disease severity.
Keywords: Later-onset; Nusinersen; SMArtCARE; Sitter; Spinal muscular atrophy.
© 2022. The Author(s).
Conflict of interest statement
A. Pechmann received compensation for advisory boards, training activities and research grants from Novartis and Biogen. C. Weiß received compensation for advisory boards, publications and/or presentations from Novartis, Biogen, and Roche. M. Baumann received compensation for advisory boards and speakers honoraria from Novartis, Biogen and Roche. M. Flotats-Bastardas received compensation for advisory boards from Biogen, Novartis and Roche. R. Trollmann reports personal fees from Desitin Pharma, Novartis, PTC Therapeutics. M. Weiler received compensation for advisory boards, consultant and speaker honoraria and/or financial support for conference attendances from Akcea Therapeutics, Alnylam Pharmaceuticals, Biogen, Pfizer, Roche, and Sobi. S. Illsinger received compensation for advisory board/consultant work from Novartis. A. Bertsche reports grants from UCB Pharma GmbH and honoraria for speaking engagements and advisory boards from Biogen GmbH, Desitin Arzneimittel GmbH, Eisai GmbH, GW Pharma GmbH, Neuraxpharm GmbH, Shire/Takeda GmbH, UCB Pharma GmbH, and ViroPharma GmbH. A. Eisenkölbl received honoraria for advisory boards, interviews and speaking engegements from Biogen, Novartis and Roche. K. Vill received speaker honoraria and travel expenses from Novartis, Biogen and Santhera. M.C. Walter received compensation for advisory boards, funding for travel or speaker honoraria, and research support from Biogen, Novartis, and Roche. J. Johannsen received honoraria for advisory board participation and/or lectures and/or manuscript writing from Avexis/Novartis, Biogen, Roche, PTC, Pfizer and Sarepta Th., respectively. C. Reihle reports travel expenses from Biogen. R. Günther received compensation for advisory boards and research support from Biogen and compensation for advisory boards from Hofmann La-Roche. T. Hagenacker received compensation for advisory boards, lectures and research grants from Novartis, Biogen and Roche. V. Horber participated in workshops and received compensation for advisory boards from Novartis and Biogen. C. Köhler received honoraria from Roche, PTC, Biogen and Genzyme. M. Müller received compensation for expert meetings and training vom Norvartis, Biogen and Roche. O. Schwarz received compensation for advisory boards and training activities and research grants from Novartis and Biogen. I. Cordts received personal fees from Biogen (speaker’s honoraria, participation in advisory boards) outside the submitted work. M. Smitka received funding for educational activities and participation in advisory boards concerning SMA from Novartis Gene Therapies, Biogen and Roche. A. Schwerin-Nagel received compensation for an advisory board from Novartis Gene Therapies. H. Kölbel received compensation for presentations and consultancy from Avexis, Biogen, Sanofi, Pfizer, Novartis, and Roche. K. Schlachter received compensation for advisory boards from Novartis, Biogen and Roche. J. Friese received compensation for an advisory board from Novartis Gene Therapies. A. Ziegler receives honoraria for advisory boards and speaker honoraria from Novartis Gene Therapies, Roche Pharma and Biogen. His institution receives research grants from Biogen. G. Bernert receives research suppert from PTC and honoraria from Novartis Gene Therapies, PTC, Biogen, Roche, Pfizer, Santhera. E. Wilichowski received compensation for advisory boards from Biogen, Novartis and Roche. H. Lochmüller received consultancy and financial support for research projects and clinical trials from Amplo Biotechnology, AMO Pharma, argenx, Biogen, Desitin, Fulcrum Therapeutics, Harmony Biosciences, KYE Pharmaceuticals, Milo Biotechnology, Novartis, Pfizer, PTC Therapeutics, Hoffman-La Roche Limited, Sanofi-Genzyme, Santhera, Sarepta, Satellos, Spark Therapeutics and Ultragenyx. HL is the Editor-in-chief for the Journal of Neuromuscular Diseases (IOS Press). J. Kirschner received support from Biogen, Novartis, Roche and ScholarRock for consultancy, educational activities and/or clinical research.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

