The Shape of the Jaw-Zebrafish Col11a1a Regulates Meckel's Cartilage Morphogenesis and Mineralization
- PMID: 36278545
- PMCID: PMC9590009
- DOI: 10.3390/jdb10040040
The Shape of the Jaw-Zebrafish Col11a1a Regulates Meckel's Cartilage Morphogenesis and Mineralization
Abstract
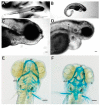
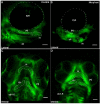
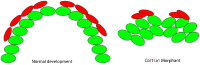
The expression of the col11a1a gene is essential for normal skeletal development, affecting both cartilage and bone. Loss of function mutations have been shown to cause abnormalities in the growth plate of long bones, as well as in craniofacial development. However, the specific effects on Meckel's cartilage have not been well studied. To further understand the effect of col11a1a gene function, we analyzed the developing jaw in zebrafish using gene knockdown by the injection of an antisense morpholino oligonucleotide using transgenic Tg(sp7:EGFP) and Tg(Fli1a:EGFP) EGFP reporter fish, as well as wildtype AB zebrafish. Our results demonstrate that zebrafish col11a1a knockdown impairs the cellular organization of Meckel's cartilage in the developing jaw and alters the bone formation that occurs adjacent to the Meckel's cartilage. These results suggest roles for Col11a1a protein in cartilage intermediates of bone development, the subsequent mineralization of the bony collar of long bones, and that which occurs adjacent to Meckel's cartilage in the developing jaw.
Keywords: Marshall syndrome; Meckel’s cartilage; Stickler syndrome type II; craniofacial; fibrochondrogenesis; jaw development; mineralization; zebrafish.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Col11a1a Expression Is Required for Zebrafish Development.J Dev Biol. 2020 Aug 28;8(3):16. doi: 10.3390/jdb8030016. J Dev Biol. 2020. PMID: 32872105 Free PMC article.
-
Diverse Fate of an Enigmatic Structure: 200 Years of Meckel's Cartilage.Front Cell Dev Biol. 2020 Aug 28;8:821. doi: 10.3389/fcell.2020.00821. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32984323 Free PMC article. Review.
-
Visualization of Chondrocyte Intercalation and Directional Proliferation via Zebrabow Clonal Cell Analysis in the Embryonic Meckel's Cartilage.J Vis Exp. 2015 Oct 21;(105):e52935. doi: 10.3791/52935. J Vis Exp. 2015. PMID: 26555721 Free PMC article.
-
gp130 is important for the normal morphogenesis of Meckel's cartilage and subsequent mandibular development.Exp Mol Med. 2007 Jun 30;39(3):295-303. doi: 10.1038/emm.2007.33. Exp Mol Med. 2007. PMID: 17603283
-
The relationship between Meckel's cartilage and the development of the human fetal mandible.Surg Radiol Anat. 1993;15(2):113-8. doi: 10.1007/BF01628309. Surg Radiol Anat. 1993. PMID: 8367789 Review.
Cited by
-
Zebrafish as a Model for Osteoporosis: Functional Validations of Genome-Wide Association Studies.Curr Osteoporos Rep. 2023 Dec;21(6):650-659. doi: 10.1007/s11914-023-00831-5. Epub 2023 Nov 16. Curr Osteoporos Rep. 2023. PMID: 37971665 Review.
-
miR-29a Is Downregulated in Progenies Derived from Chronically Stressed Males.Int J Mol Sci. 2023 Sep 14;24(18):14107. doi: 10.3390/ijms241814107. Int J Mol Sci. 2023. PMID: 37762407 Free PMC article.
-
Early stress exposure on zebrafish development: effects on survival, malformations and molecular alterations.Fish Physiol Biochem. 2024 Aug;50(4):1545-1562. doi: 10.1007/s10695-024-01355-0. Epub 2024 May 14. Fish Physiol Biochem. 2024. PMID: 38743196 Free PMC article.
References
-
- Kimmel C., Miller C., Kruze G., Ullmann B., BreMiller R., Larison K., Snyder H. The shaping of pharyngeal cartilages during early development of the zebrafish. Dev. Biol. 1998;203:245–263. - PubMed
-
- Schilling T., Kimmel C. Segment and cell type lineage restrictions during pharyngeal arch development in the zebrafish embryo. Development. 1994;120:483–494. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

