Remote Eradication of Bacteria on Orthopedic Implants via Delayed Delivery of Polycaprolactone Stabilized Polyvinylpyrrolidone Iodine
- PMID: 36278664
- PMCID: PMC9589933
- DOI: 10.3390/jfb13040195
Remote Eradication of Bacteria on Orthopedic Implants via Delayed Delivery of Polycaprolactone Stabilized Polyvinylpyrrolidone Iodine
Abstract
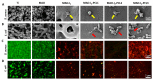
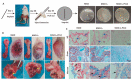
Bacteria-associated late infection of the orthopedic devices would further lead to the failure of the implantation. However, present ordinary antimicrobial strategies usually deal with early infection but fail to combat the late infection of the implants due to the burst release of the antibiotics. Thus, to fabricate long-term antimicrobial (early antibacterial, late antibacterial) orthopedic implants is essential to address this issue. Herein, we developed a sophisticated MAO-I2-PCLx coating system incorporating an underlying iodine layer and an upper layer of polycaprolactone (PCL)-controlled coating, which could effectively eradicate the late bacterial infection throughout the implantation. Firstly, micro-arc oxidation was used to form a microarray tubular structure on the surface of the implants, laying the foundation for iodine loading and PCL bonding. Secondly, electrophoresis was applied to load iodine in the tubular structure as an efficient bactericidal agent. Finally, the surface-bonded PCL coating acts as a controller to regulate the release of iodine. The hybrid coatings displayed great stability and control release capacity. Excellent antibacterial ability was validated at 30 days post-implantation via in vitro experiments and in vivo rat osteomyelitis model. Expectedly, it can become a promising bench-to-bedside strategy for current infection challenges in the orthopedic field.
Keywords: antibacterial coating; micro-arc oxidation; orthopedic implants; polycaprolactone; povidone-iodine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
A trilogy antimicrobial strategy for multiple infections of orthopedic implants throughout their life cycle.Bioact Mater. 2020 Dec 10;6(7):1853-1866. doi: 10.1016/j.bioactmat.2020.11.030. eCollection 2021 Jul. Bioact Mater. 2020. PMID: 33336116 Free PMC article.
-
In vitro and in vivo evaluation of structurally-controlled silk fibroin coatings for orthopedic infection and in-situ osteogenesis.Acta Biomater. 2020 Oct 15;116:223-245. doi: 10.1016/j.actbio.2020.08.040. Epub 2020 Sep 2. Acta Biomater. 2020. PMID: 32889111
-
Iodine Immobilized Metal-Organic Framework for NIR-Triggered Antibacterial Therapy on Orthopedic Implants.Small. 2021 Sep;17(35):e2102315. doi: 10.1002/smll.202102315. Epub 2021 Jul 26. Small. 2021. PMID: 34309186
-
Surface engineering of biomaterials in orthopedic and dental implants: Strategies to improve osteointegration, bacteriostatic and bactericidal activities.Biotechnol J. 2021 Jul;16(7):e2000116. doi: 10.1002/biot.202000116. Epub 2021 Apr 17. Biotechnol J. 2021. PMID: 33813785 Review.
-
Surface treatment strategies to combat implant-related infection from the beginning.J Orthop Translat. 2018 Sep 28;17:42-54. doi: 10.1016/j.jot.2018.09.001. eCollection 2019 Apr. J Orthop Translat. 2018. PMID: 31194031 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

