Challenges and Possibilities of Cell-Based Tissue-Engineered Vascular Grafts
- PMID: 36285145
- PMCID: PMC9494692
- DOI: 10.34133/2021/1532103
Challenges and Possibilities of Cell-Based Tissue-Engineered Vascular Grafts
Abstract
There is urgent demand for biologically compatible vascular grafts for both adult and pediatric patients. The utility of conventional nonbiodegradable materials is limited because of their thrombogenicity and inability to grow, while autologous vascular grafts involve considerable disadvantages, including the invasive procedures required to obtain these healthy vessels from patients and insufficient availability in patients with systemic atherosclerosis. All of these issues could be overcome by tissue-engineered vascular grafts (TEVGs). A large body of evidence has recently emerged in support of TEVG technologies, introducing diverse cell sources (e.g., somatic cells and stem cells) and novel fabrication methods (e.g., scaffold-guided and self-assembled approaches). Before TEVG can be applied in a clinical setting, however, several aspects of the technology must be improved, such as the feasibility of obtaining cells, their biocompatibility and mechanical properties, and the time needed for fabrication, while the safety of supplemented materials, the patency and nonthrombogenicity of TEVGs, their growth potential, and the long-term influence of implanted TEVGs in the body must be assessed. Although recent advances in TEVG fabrication have yielded promising results, more research is needed to achieve the most feasible methods for generating optimal TEVGs. This article reviews multiple aspects of TEVG fabrication, including mechanical requirements, extracellular matrix components, cell sources, and tissue engineering approaches. The potential of periodic hydrostatic pressurization in the production of scaffold-free TEVGs with optimal elasticity and stiffness is also discussed. In the future, the integration of multiple technologies is expected to enable improved TEVG performance.
Copyright © 2021 Junichi Saito et al.
Conflict of interest statement
The authors declare no competing interests.
Figures

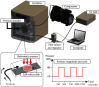
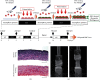
Similar articles
-
Preclinical study of patient-specific cell-free nanofiber tissue-engineered vascular grafts using 3-dimensional printing in a sheep model.J Thorac Cardiovasc Surg. 2017 Apr;153(4):924-932. doi: 10.1016/j.jtcvs.2016.10.066. Epub 2016 Nov 14. J Thorac Cardiovasc Surg. 2017. PMID: 27938900 Free PMC article.
-
Bioresorbable silk grafts for small diameter vascular tissue engineering applications: In vitro and in vivo functional analysis.Acta Biomater. 2020 Mar 15;105:146-158. doi: 10.1016/j.actbio.2020.01.020. Epub 2020 Jan 17. Acta Biomater. 2020. PMID: 31958596 Free PMC article.
-
Hydrostatic pressure under hypoxia facilitates fabrication of tissue-engineered vascular grafts derived from human vascular smooth muscle cells in vitro.Acta Biomater. 2023 Nov;171:209-222. doi: 10.1016/j.actbio.2023.09.041. Epub 2023 Oct 2. Acta Biomater. 2023. PMID: 37793599
-
Tissue engineered vascular grafts: current state of the field.Expert Rev Med Devices. 2017 May;14(5):383-392. doi: 10.1080/17434440.2017.1324293. Epub 2017 May 9. Expert Rev Med Devices. 2017. PMID: 28447487 Review.
-
Tissue engineered small-diameter vascular grafts.Clin Plast Surg. 2003 Oct;30(4):507-17. doi: 10.1016/s0094-1298(03)00069-5. Clin Plast Surg. 2003. PMID: 14621299 Review.
Cited by
-
Vascular Grafts: Technology Success/Technology Failure.BME Front. 2023 Jan 16;4:0003. doi: 10.34133/bmef.0003. eCollection 2023. BME Front. 2023. PMID: 37849668 Free PMC article. Review.
-
Potential of Autologous Progenitor Cells and Decellularized Porcine Artery Matrix in Construction of Tissue-engineered Vascular Grafts.Organogenesis. 2021 Oct 2;17(3-4):72-84. doi: 10.1080/15476278.2021.1963603. Epub 2021 Aug 18. Organogenesis. 2021. PMID: 34405770 Free PMC article.
-
In Silico Trials of Prosthetic Valves Replicate Methodologies for Evaluating the Fatigue Life of Artificial Leaflets to Expand Beyond In Vitro Tests and Conventional Clinical Trials.Biomedicines. 2025 May 7;13(5):1135. doi: 10.3390/biomedicines13051135. Biomedicines. 2025. PMID: 40426962 Free PMC article.
-
Light-based 3D bioprinting techniques for illuminating the advances of vascular tissue engineering.Mater Today Bio. 2024 Oct 2;29:101286. doi: 10.1016/j.mtbio.2024.101286. eCollection 2024 Dec. Mater Today Bio. 2024. PMID: 39435375 Free PMC article. Review.
-
Mitigating challenges and expanding the future of vascular tissue engineering-are we there yet?Front Physiol. 2023 Jan 4;13:1079421. doi: 10.3389/fphys.2022.1079421. eCollection 2022. Front Physiol. 2023. PMID: 36685187 Free PMC article. No abstract available.
References
Publication types
LinkOut - more resources
Full Text Sources

