LC3-dependent EV loading and secretion (LDELS) promotes TFRC (transferrin receptor) secretion via extracellular vesicles
- PMID: 36286616
- PMCID: PMC10240981
- DOI: 10.1080/15548627.2022.2140557
LC3-dependent EV loading and secretion (LDELS) promotes TFRC (transferrin receptor) secretion via extracellular vesicles
Abstract
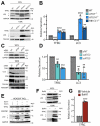
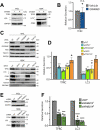
LC3-dependent EV loading and secretion (LDELS) is a secretory autophagy pathway in which the macroautophagy/autophagy machinery facilitates the packaging of cytosolic cargos, such as RNA-binding proteins, into extracellular vesicles (EVs) for secretion outside of the cell. Here, we identify TFRC (transferrin receptor), one of the first proteins found to be secreted via EVs, as a transmembrane cargo of the LDELS pathway. Similar to other LDELS targets, TFRC secretion via EVs genetically requires components of the MAP1LC3/LC3-conjugation machinery but is independent of other ATGs involved in classical autophagosome formation. Furthermore, the packaging and secretion of this transmembrane protein into EVs depends on multiple ESCRT pathway components and the small GTPase RAB27A. Based on these results, we propose that the LDELS pathway promotes TFRC incorporation into EVs and its secretion outside the cell.Abbreviations: ATG: autophagy related; ESCRT: endosomal sorting complexes required for transport; EV: extracellular vesicle; EVP: extracellular vesicle and particle; ILV: intralumenal vesicle; LDELS: LC3-dependent EV loading and secretion; LIR: LC3-interacting region; MVE: multivesicular endosome; RBP: RNA-binding protein; TMT: tandem mass tag; TFRC: transferrin receptor.
Keywords: ATG7; ATG8; LC3-conjugation; extracellular vesicles; secretory autophagy; transferrin receptor.
Conflict of interest statement
JD is a member of the Scientific Advisory Board of Vescor Therapeutics, LLC.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
