Combining stem cell rejuvenation and senescence targeting to synergistically extend lifespan
- PMID: 36287172
- PMCID: PMC9648810
- DOI: 10.18632/aging.204347
Combining stem cell rejuvenation and senescence targeting to synergistically extend lifespan
Abstract
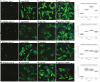
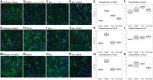
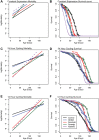
Why biological age is a major risk factor for many of the most important human diseases remains mysterious. We know that as organisms age, stem cell pools are exhausted while senescent cells progressively accumulate. Independently, induction of pluripotency via expression of Yamanaka factors (Oct4, Klf4, Sox2, c-Myc; OKSM) and clearance of senescent cells have each been shown to ameliorate cellular and physiological aspects of aging, suggesting that both processes are drivers of organismal aging. But stem cell exhaustion and cellular senescence likely interact in the etiology and progression of age-dependent diseases because both undermine tissue and organ homeostasis in different if not complementary ways. Here, we combine transient cellular reprogramming (stem cell rejuvenation) with targeted removal of senescent cells to test the hypothesis that simultaneously targeting both cell-fate based aging mechanisms will maximize life and health span benefits. We find that OKSM extends lifespan and show that both interventions protect the intestinal stem cell pool, lower inflammation, activate pro-stem cell signaling pathways, and synergistically improve health and lifespan. Our findings suggest that a combination therapy, simultaneously replacing lost stem cells and removing senescent cells, shows synergistic potential for anti-aging treatments. Our finding that transient expression of both is the most effective suggests that drug-based treatments in non-genetically tractable organisms will likely be the most translatable.
Keywords: aging; senescence; stem cells.
Conflict of interest statement
Figures



Similar articles
-
Rejuvenating aged stem cells: therapeutic strategies to extend health and lifespan.FEBS Lett. 2024 Nov;598(22):2776-2787. doi: 10.1002/1873-3468.14865. Epub 2024 Apr 11. FEBS Lett. 2024. PMID: 38604982 Free PMC article. Review.
-
Transient non-integrative expression of nuclear reprogramming factors promotes multifaceted amelioration of aging in human cells.Nat Commun. 2020 Mar 24;11(1):1545. doi: 10.1038/s41467-020-15174-3. Nat Commun. 2020. PMID: 32210226 Free PMC article.
-
Turning back time with emerging rejuvenation strategies.Nat Cell Biol. 2019 Jan;21(1):32-43. doi: 10.1038/s41556-018-0206-0. Epub 2019 Jan 2. Nat Cell Biol. 2019. PMID: 30602763 Free PMC article. Review.
-
Nucleostemin rejuvenates cardiac progenitor cells and antagonizes myocardial aging.J Am Coll Cardiol. 2015 Jan 20;65(2):133-47. doi: 10.1016/j.jacc.2014.09.086. J Am Coll Cardiol. 2015. PMID: 25593054 Free PMC article.
-
The bright and dark side of skin senescence. Could skin rejuvenation anti-senescence interventions become a "bright" new strategy for the prevention of age-related skin pathologies?Mech Ageing Dev. 2021 Jan;193:111409. doi: 10.1016/j.mad.2020.111409. Epub 2020 Nov 26. Mech Ageing Dev. 2021. PMID: 33249190 Review.
Cited by
-
Mechanisms Underlying Brain Aging Under Normal and Pathological Conditions.Neurosci Bull. 2023 Feb;39(2):303-314. doi: 10.1007/s12264-022-00969-9. Epub 2022 Nov 27. Neurosci Bull. 2023. PMID: 36437436 Free PMC article. Review.
-
Cancer progression through the lens of age-induced metabolic reprogramming.Nat Rev Cancer. 2025 Jul 11. doi: 10.1038/s41568-025-00845-4. Online ahead of print. Nat Rev Cancer. 2025. PMID: 40646271 Review.
-
SASP Modulation for Cellular Rejuvenation and Tissue Homeostasis: Therapeutic Strategies and Molecular Insights.Cells. 2025 Apr 17;14(8):608. doi: 10.3390/cells14080608. Cells. 2025. PMID: 40277933 Free PMC article. Review.
-
Targeting multiple hallmarks of mammalian aging with combinations of interventions.Aging (Albany NY). 2024 Aug 18;16(16):12073-12100. doi: 10.18632/aging.206078. Epub 2024 Aug 18. Aging (Albany NY). 2024. PMID: 39159129 Free PMC article. Review.
-
Therapeutic targeting of senescent cells in the CNS.Nat Rev Drug Discov. 2024 Nov;23(11):817-837. doi: 10.1038/s41573-024-01033-z. Epub 2024 Sep 30. Nat Rev Drug Discov. 2024. PMID: 39349637 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

