Glucose/Fructose Delivery to the Distal Nephron Activates the Sodium-Chloride Cotransporter via the Calcium-Sensing Receptor
- PMID: 36288902
- PMCID: PMC10101570
- DOI: 10.1681/ASN.2021121544
Glucose/Fructose Delivery to the Distal Nephron Activates the Sodium-Chloride Cotransporter via the Calcium-Sensing Receptor
Abstract
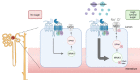
Background: The calcium-sensing receptor (CaSR) in the distal convoluted tubule (DCT) activates the NaCl cotransporter (NCC). Glucose acts as a positive allosteric modulator of the CaSR. Under physiologic conditions, no glucose is delivered to the DCT, and fructose delivery depends on consumption. We hypothesized that glucose/fructose delivery to the DCT modulates the CaSR in a positive allosteric way, activating the WNK4-SPAK-NCC pathway and thus increasing salt retention.
Methods: We evaluated the effect of glucose/fructose arrival to the distal nephron on the CaSR-WNK4-SPAK-NCC pathway using HEK-293 cells, C57BL/6 and WNK4-knockout mice, ex vivo perfused kidneys, and healthy humans.
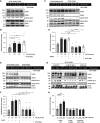
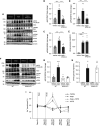
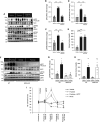
Results: HEK-293 cells exposed to glucose/fructose increased SPAK phosphorylation in a WNK4- and CaSR-dependent manner. C57BL/6 mice exposed to fructose or a single dose of dapagliflozin to induce transient glycosuria showed increased activity of the WNK4-SPAK-NCC pathway. The calcilytic NPS2143 ameliorated this effect, which was not observed in WNK4-KO mice. C57BL/6 mice treated with fructose or dapagliflozin showed markedly increased natriuresis after thiazide challenge. Ex vivo rat kidney perfused with glucose above the physiologic threshold levels for proximal reabsorption showed increased NCC and SPAK phosphorylation. NPS2143 prevented this effect. In healthy volunteers, cinacalcet administration, fructose intake, or a single dose of dapagliflozin increased SPAK and NCC phosphorylation in urinary extracellular vesicles.
Conclusions: Glycosuria or fructosuria was associated with increased NCC, SPAK, and WNK4 phosphorylation in a CaSR-dependent manner.
Copyright © 2022 by the American Society of Nephrology.
Conflict of interest statement
I. Alquisiras-Burgos reports receiving research funding
Figures




Comment in
-
Sweet-Talking the Distal Nephron Calcium-Sensing Receptor.J Am Soc Nephrol. 2023 Jan 1;34(1):1-2. doi: 10.1681/ASN.2022091054. Epub 2022 Oct 26. J Am Soc Nephrol. 2023. PMID: 36288903 Free PMC article. No abstract available.
References
-
- Gamba G: The evolving field of salt transport regulation in the Steve Hebert Lecture. Am J Physiol Renal Physiol 311: F68–F70, 2016 - PubMed
-
- Simon DB, Nelson-Williams C, Bia MJ, Ellison D, Karet FE, Molina AM, et al. : Gitelman’s variant of Bartter’s syndrome, inherited hypokalaemic alkalosis, is caused by mutations in the thiazide-sensitive Na-Cl cotransporter. Nat Genet 12: 24–30, 1996 - PubMed
-
- Lifton RP, Gharavi AG, Geller DS: Molecular mechanisms of human hypertension. Cell 104: 545–556, 2001 - PubMed
-
- Louis-Dit-Picard H Barc J Trujillano D Miserey-Lenkei S Bouatia-Naji N Pylypenko O et al. ; International Consortium for Blood Pressure (ICBP) : KLHL3 mutations cause familial hyperkalemic hypertension by impairing ion transport in the distal nephron. Nat Genet 44: 456–460, S1–S3, 2012 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

